NNadir
NNadir's JournalPalladium is a fission product.
The paper I will discuss in this very brief post is this one: Enhancement of Thermoelectric Properties in Pd–In Co-Doped SnTe and Its Phase Transition Behavior. (Wang et al ACS Appl. Mater. Interfaces 2019 11 37 33792-33802)
I don't have time this morning for much more than this.
Thermoelectric devices are well known and have been utilized, most famously in the space program, for the direct conversion of heat into electricity.
As an advocate of the last, best hope of saving the planet, nuclear energy, I follow the science around these devices rather closely.
The historical thermoelectric devices, including those that powered the Voyager spacecraft - with which we are still in contact after many decades - had low energy efficiency: Typically the heat energy converted to electrical energy was less than 10%. The efficiency of these devices is described in science and engineering by a parameter known as the Seeback coefficient, usually referred to by the somewhat confusing symbol "ZT" which is not an algebraic product.
I consider that all of the components of so called "nuclear waste" are valuable and need to be recovered, both as useful radioactive materials and as non-radioactive materials.
It is possible to isolate non-radioactive palladium from fission products in the case where fast separation of ruthenium is undertaken, since ruthenium-106 has a relatively short half-life, roughly a year. This is possible for fluid phased reactors of a type that has been popularly discussed among modern nuclear engineers. Ru-106 decays to stable palladium-106.
However the isolation of palladium from used nuclear fuel that has been foolishly allowed to accumulate without being put to use will contain a long lived radioactive isotope, Pd-107, which decays with a long half life to stable Ag-107. I believe that this palladium is also useful and that the risk associated with its radioactivity is vanishingly small if it is utilized in devices, which is why this paper caught my eye.
In the fast fission of plutonium - which in my view is the key to making nuclear fuel inexhaustible - about 6% of the fission products are palladium. (Natural palladium contains the 102 isotope which is observationally stable but probably radioactive with an a half-life so long as to be undetectable. Used nuclear fuel will contain this mass number in the form of stable ruthenium-102)
Here is figure 10 from the paper:

Here is the caption:
From the conclusion of the paper:
This is a very high ZT value. To save the world from climate change, it is necessary to utilize high temperature nuclear reactors with high thermal efficiency in a combined cycle fashion, with some of the energy being utilized to make chemical fuels that can be obtained from hydrogen and carbon oxides, both CO and CO2. CO2 can also be reduced to carbon for use in materials, which is essentially combustion in reverse.
Temperatures of 800 K are readily accessible under these conditions, and may be utilized in the cooling phases necessary in the thermochemical conversion of carbon oxides to economically viable closed cycles approaches to carbon utilization.
It's cool I think.
None of this is likely to happen by the way, since fear and ignorance are obviously triumphant in modern times, but it is feasible that ignorance will fail, both on the right, where it is politically popular, and on the left, where anti-nuclear stupidity is popular.
I hope you will have a wonderful weekend.
Carbocations Generated Electrochemically from Carboxylic Acids.
The paper I'll discuss in this post is this one: Hindered dialkyl ether synthesis with electrogenerated carbocations (Baran et al, Nature 573, 398–402 (2019)).
Last year my kid was in France where he was working with some organosilanes and he asked me to scan and email an organic chemistry textbook and some problem solutions so he could teach himself some organic chemistry. He told me he was thinking of taking some organic chemistry courses for one of the minors he was considering.
I said, "Well that's OK with me, but if you take courses in organic chemistry, just don't fall in love with it."
Medicinal chemistry is a dying field in the United States, and with it, the economic value of degrees in chemistry focusing on organic chemistry. That's really terrible, since organic chemistry is such a beautiful science and the history of organic chemistry in the United States is sublime.
It's been some decades since I've worked in the field, but I recall how very much alive I felt in those days, young, newly married to the woman of my dreams and still going to work on the weekends not because I had to do so, not because anyone was paying me to do so, but simply because I loved doing it.
I suppose on a certain level a knowledge of organic chemistry still permeates my day to day life and work, but it's peripheral; I'm sure I've forgotten more than I know.
But today I came across this beautiful and in someways earth shattering beautiful little paper in a major cross disciplinary scientific journal where one doesn't see that much organic synthesis anymore, Nature. (There is a Nature Journal called Nature Chemistry, but I don't read it all that much unless some reference drags me there.)
From the introduction:
I haven't kept up on things, but right off the bat that strikes me as a big deal, carbocations from simple carboxylic acids.
Here's the stuff from the world in which I used to live:
The Mitsunobu reaction is always a favorite, since it involves a reagent known as DEAD, (diethyl azodicarboxylate).
The pictures tell the story:
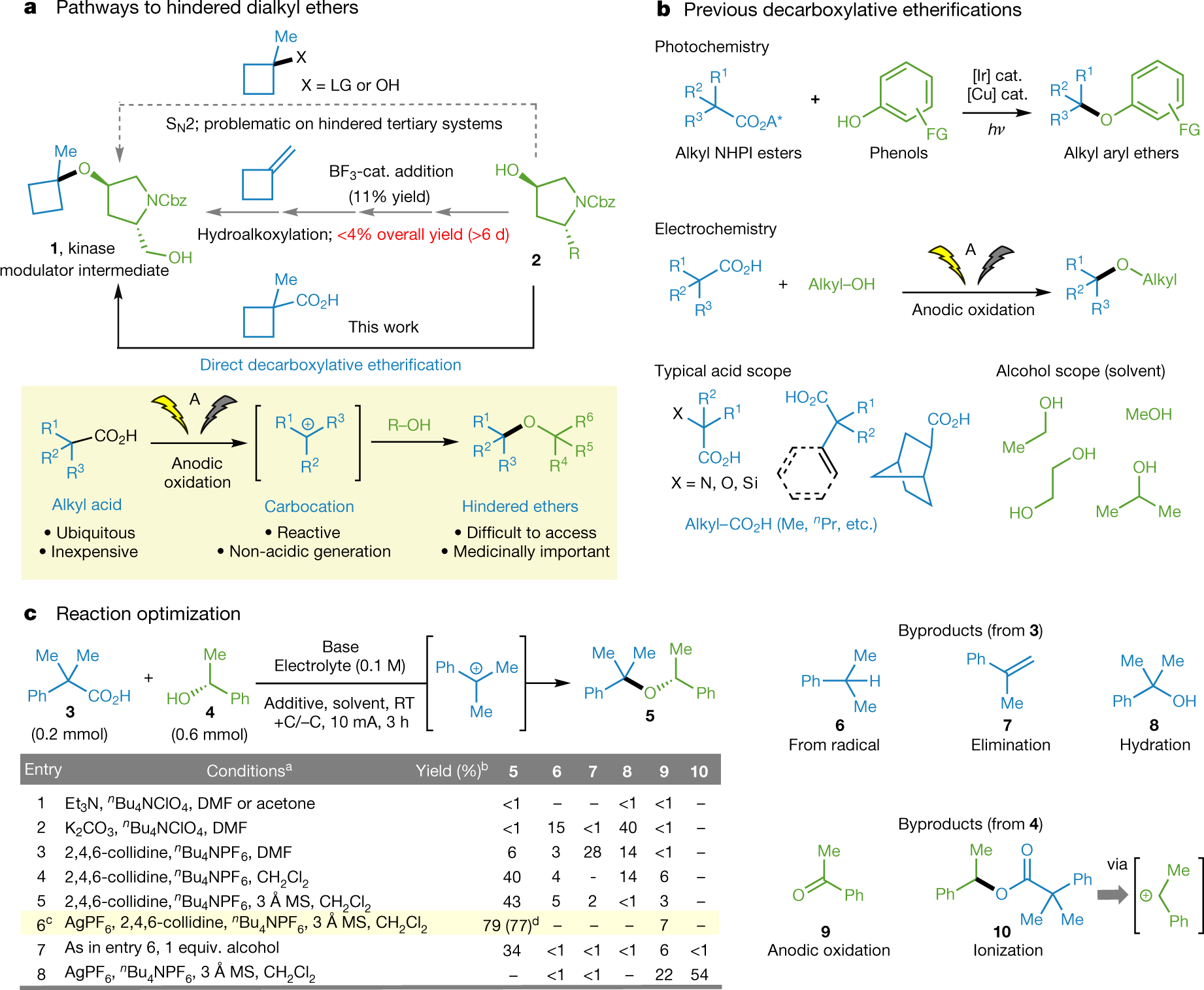
The caption:
Actually, this chemistry is quite old according to the authors:
It's chemistry with which I'm unfamiliar, for some reason I've always looked askance at electrochemistry, at least until recently, when I have come to regret my intellectual laziness in this regard.
Some structures they synthesized:

The caption:
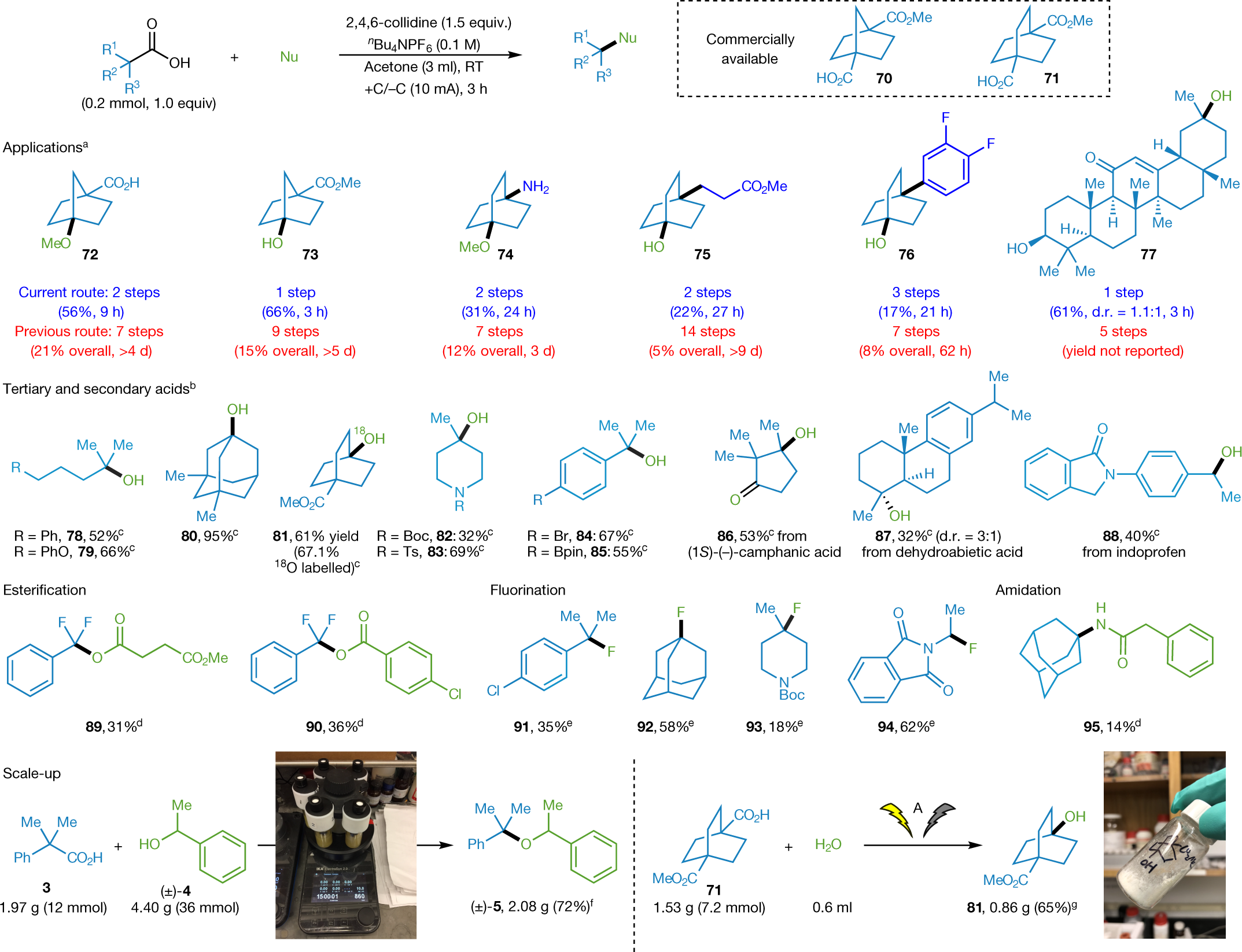
The caption:
The conclusion of the paper strikes me as something of an understatement:
This is by the way, molten salt chemistry, in the extreme, since a key reagent is nBu4NPF6. Electrochemistry in molten salts would be a very big deal in a sane world, not the one we live in, a world where organic chemistry would regain some value.
I know, I know, I know...it's very esoteric...very esoteric...
It's very esoteric, but in a time of so much tragedy, children in cages, a destructive nutcase running the country, it is well worth finding some peace in the knowledge that there still are wonderful and beautiful things left, even in this country.
I hope you will enjoy a very pleasant up coming weekend.
Structural basis of nucleosome recognition and modification by MLL methyltransferases
The paper I'll discuss in this post has the title I've used for the post itself: Structural basis of nucleosome recognition and modification by MLL methyltransferases (Jing Huang et al Nature 573, 445–449 (2019))
Last night I had the pleasure of attending a lecture by Dr. Benjamin Garcia who is a world expert in the structure of chromatin, specifically, the epigenetic implications of particular post translational modifications of histones. Histones are the proteins that wrap DNA, choreographing the way they and the genes that constitute them turn on and off, to simplify the matter somewhat. The histones - there are four of them - are extremely basic proteins, inasmuch as they are rich in arginine and lysine, and it is the chemistry of the latter amino acid, lysine, that is a controlling factor in how histones behave and function.
(As aside, the evening was an embarrassment of riches, Dr. Garcia's lecture was followed by one by Dr. Vicki Wysocki - in the picture of her lab group she is in the back row on the far left, partially obscured by one of her students. Dr. Wysocki is a world leader in the use of mass spectrometry to study protein complex structures, that is how proteins interact with one another to conduct the business of metabolism. She showed work where she defined the structure of an hexameric protein by mass spectrometry that was later confirmed by cryo EM imagining (the imaging was problematic so her group took a shot at it by mass spec) - no small trick.)
Anyway, about histones: As they control the operation of DNA, they are obviously involved in many processes involving cell division, both normal cell division and abnormal cell division, notably cancer. It is known that specific residues, in particular the ?-amino group in lysine, in the protein sequences of the histones are modified generally (but not always) in one of two ways, by acetylation or methylation. A bit of nomenclature: The term "H3K4" refers to histone 3, "H3" having a lysine residue (K in peptide language) in the 4 position in the amino acid sequence. The term "H2BK120" refers to histone 2B's lysine in the 120th residue of the sequence.
The histones in turn can also choreograph or allow the chemical modification of DNA itself - DNA can be methylated - this is the area of "epigenetics" which controls many areas of cell function and behavior, including, it seems, aging.
I've played in this space professionally. It's fascinating.
Before producing excerpts of the texts, it is probably useful to produce a graphic from the paper showing what this chromatin complex of histones and DNA looks like:

The caption:
The abstract and introduction describes what some of these abbreviations mean:
This is a paper about the epigenetics of leukemia.
The "complexes" here are what Dr. Wysocki's group studies, not by imaging (Cryo-EM) but be interference from mass spectrometry data. Very cool.
Anyway, some more text.
RBBP5 is a retinoblastoma binding protein that is important in cell division; it is a tumor suppressor gene. (One of the interesting points of Dr. Garcia's talk last night concerned the interaction of histone tails with protein sequences that seem to be involved in immune function - the immunology of cancer is another interesting topic.)
I recognize that this is all very esoteric, and I produce it because of the high the lectures gave me, so let me limit the rest of this post to some pretty pictures of chromatin histone/DNA complexes with captions.

The caption:

The caption:
 ?as=webp
?as=webp
The caption:
From the paper's conclusion:
Well, to each his own; I guess you had to be there.
Even cancer can have its own magnificence.
Evenings like last evening make my life worth living, such beautiful science albeit in a sadly dying world.
I hope your work week has had as many moments of transcendent joy as mine has had.
Electricity Prices Around the World 2017

Source: IEA Electricity Information 2018, Page IV.8
An Interactive Map of Severely PFAS Contaminated Sites in the US.
These are the perfluoralkanoates, widely used persistent organic chemicals where all, or most of the hydrogens have been replaced with fluorine. It appears that their physiology of these compounds is problematic.
There are probably no human beings who are not contaminated with them at some level, but some are far more contaminated than others. (The dose makes the poison.)
I was able to click on sites near my home, and found that a small commercial airport for commuter traffic and served by at least one commuter airline was a former military base that has profound PFAS contamination.
https://www.ewg.org/interactive-maps/2019_pfas_contamination/map/
Today's Google Doodle Honors an Important and Unsung Type of Scientist, the "Amateur" Scientist.
There are scientists, very important scientists in fact, who do their science as something other as a job. Some of these are autodidacts, who teach themselves what they need to know and go on to advance their fields greatly, often in obscurity that outlives their physical lives, with fame coming after their deaths.
These kinds of people are invaluable.
Gregor Mendel was such a scientist; his job was "Monk," but he did science that survived him and helped pave the way for modern genetics.
Today's Google Doodle is about another such scientist: Ynes Mexia
I personally love the Google Doodles; they teach me things I didn't know.
Electricity Prices Around the World 2013.
Excuse this experiment in software management. I am working to gain better use of graphics in blog posts as I consider starting a private blog of my own.
This test graphic comes from an article I've been writing for some time on why negative electricity prices are economically and environmentally destructive. It will contain this graphic from a report by the interesting energy thinker at MIT, whose work I follow closely, Charles Forsberg. For the record, he is not nearly as hostile to so called "renewable energy" as I am - I think of it as an expensive sacred cow that is speeding the destruction of the planet - but despite this disagreement, he is an important realistic thinker on decarbonization with broad multidisciplinary insights.

The full report is here: MIT-ANP-TR-162
This post is just a test, and if it annoys anyone, I apologize.
It's unusual, but a pundit actually cause me to change my sig line here.
I have no use for pundits, or for journalists in general, since they have played too large a role in normalizing the reprehensible.
But I came across some punditry that was actually, in my cynical view, quite thoughtful in its appeal to FDR, and took a quote from it as my new sig line.
The pundit's remarks, from CNN, are here: The History Lesson 2020 Democrats Cannot Afford to Ignore.
The text therein is my new sig line for the time being. (I do change it frequently.)
Genetic Sequencing of Mahi Mahi to Determine the Exposure Level From the Deep Horizon Oil Disaster.
The paper I'll discuss in this relatively brief post is this one: Whole-Transcriptome Sequencing of Epidermal Mucus as a Novel Method for Oil Exposure Assessment in Juvenile Mahi-Mahi (Coryphaena hippurus) (Justin B. Greer,*,† Nicolette E. Andrzejczyk,*,† Edward M. Mager,‡ John D. Stieglitz,§ Daniel Benetti,§ Martin Grosell,∥ and Daniel Schlenk†,?, Environ. Sci. Technol. Lett. 2019, 6, 538?544)
One of the joys of writing posts in this "sleepy little DU science forum" - as I've heard it described - is the privilege of learning things as I write. Over the last few weeks, I've been writing a somewhat involved post comparing two recently published scientific papers about two radioactive contamination events, one of which is everyone's favorite with the possible exception of Chernobyl, Fukushima, and the other involving radioactive contamination of a San Joaquin oil field in California. Writing this rather long post has stimulated me to do some interesting reading on the human physiology of certain radioactive nuclides, including two that surely killed a rather famous scientist.
One of the side notes I found myself going down is the case of the famous scientific paper about the "Fukushima Tuna Fish" which received vast international attention, much to the chagrin of the paper's authors, and believe me, when a scientist gets a paper with an international attention all over the news media, "chagrin" is not usually the word associated with the attention.
Anyway, this is not that post. I'm still working on it and it isn't done.
It proves to timely: The recent Fukushima attention concerns the proposal to dump "radioactive" seawater into the sea - it happens that there is no such thing, and never has been such a thing as seawater that isn't radioactive - the dumping is perfectly OK with me. If one supports nuclear energy as I do, one has to greet such ignorance with a mixture of amusement and despair. If the Fukushima "radioactive seawater" is dumped, we can expect the morons at Greenpeace to have a festival of clownish trivializing stupidity featuring dressing up and cruising around in diesel powered boats in their ongoing efforts to be sure the planet commits a suicide worthy of the Darwin Award.
I'm sure the reactions will be as stupid as the reaction I experienced here some time ago when a correspondent dug up one of my old posts to announce that the world was ending because a tunnel collapsed on the radioactively contaminated Hanford Nuclear Weapons Complex in Washington State, a collapse also widely reported in the media, albeit not as actively as the famous Fukushima tuna fish.
God bless the ignore list.
Anyway, the paper here is about another kind of fish, a fish contaminated by petroleum leaks, in this case, oil from the Deepwater Horizon Oil.
From the introduction to the paper:
As biological barriers go, the one with which I am most familiar is human skin, with particular focus on its molecular biology, human skin a fascinating organ, but the authors here are concerned with another complex biological barrier, the mucus layer coating fish, also a fascinating organ with fascinating molecular biology which, as the authors note...
The authors focus on an usually studied subclass of biological barrier molecules, specifically RNA.
Recently there has been increased interest in what is called the "exposome" which is the molecular signatures of exposure to external molecules and stresses, many of which ultimately show up in toxicological syndromes. This is a relatively new undertaking and is proving to be quite fascinating. Here, for example is a link to a recent paper upon which I stumbled, the expsome associated with Alzheimer's disease: The Alzheimer's Disease Exposome. Publication of this paper, by the way, does not "prove" anything at all about Alzheimer's disease, but it offers an important area of inquiry worthy of study.
As they are sometimes and somewhat naively interpreted, the nucleic acids represent something like a computer program of life, and these "programs" are of two types, the germline type - that which is heritable - and the somatic type, that which is involved in the control and feedback loops of the molecular biology of living things. In somatic nucleic acids, a number of changes accumulate normally during life, these are called "epigenetic changes" and as such represent the exposome. These somatic changes are known to control normal and abnormal processes like, in the former case, aging and development, in the latter case, diseases like cancer and autoimmune diseases like lupus and rheumatoid arthritis.
The oil utilized in this study to examine the effects of Deepwater Horizon spill was real Deepwater Horizon Oil, collected from the sea and stored as such:
Cool, I think. I'm glad scientists collected "DWH" oil samples and stored them for study years after the fact. Excellent scientific forethought!
Some graphics from the paper:


The authors studied, using modern technology, 194,282 fish genes in the mucus barrier.
They found:
... Together, our data suggest that mucosal mRNA abundance may be indicative of whole-animal changes in immune-related function in PAH-exposed fish. IPA also predicted alterations in toxicity functions induced by oil exposure. The top-ranked toxicity functions in the low- HEWAF exposure were liver fibrosis, liver hyperplasia, congenital heart anomaly, and cardiac enlargement (Figure 1C and Table S12). Similarly, liver enlargement, pulmonary hypertension, liver hyperbilirubinemia, and liver hyperplasia were the top-ranked toxicity functions in the high-HWAF exposure. Of all toxicity functions, 17 cardiac functions were predicted to be altered in the low-HEWAF exposure and 11 in the high-HEWAF exposure, with many overlapping categories such as cardiac enlargement, cardiac arrhythmia, cardiac fibrosis, and cardiac necrosis/cell death (Table S12). In addition, there was a predicted inhibition of cardiac muscle function, cardiac muscle contractility, and abnormality of the heart ventricle (Figure 2B), which are cardiac phenotypes known to be altered by crude oil-derived PAH exposure in fish.37,38 Alterations in Ca2+ homeostasis were also predicted from the mucus transcriptional profile, such as decreased mobilization of Ca2+ and an increased quantity of Ca2+ (Figure 2B). Transcripts of ryanodine receptor 2 (ryr2), the primary mediator of calcium-induced Ca2+ release required for cardiomyocyte contraction, were upregulated in the mucus and have also been shown to be dysregulated in oil-exposed mahi-mahi and Atlantic haddock (Melanogrammus aeglef inus) embryos (Table S11).39,40
IPA here refers to "Ingenuity Pathway Analysis" a bioinformatics software tool. IPA, Qiagen
The authors state that to their knowledge, this is the first paper to look at this particular pathway in determining the exposome of oil spills on marine life.
This paper will get no attention from our distracted media with its selective attention, the same media that gave the intellectually and morally challenged awful criminal Donald J. Trump the White House, "...because...her emails..."
In reality the "election" of Donald J. Trump is a trivial, if wholly unfortunate, blip in world history. The destruction of the planetary atmosphere and oceans by dangerous fossil fuels like, but not limited to, petroleum, "...because...Fukushima..." is not trivial.
I wish you a pleasant and safe Friday the 13th.
Unexpectedly Increased Particle Emissions from the Steel Industry Using Desulfurization Technology.
The paper I'll discuss briefly in this post is this one: Unexpectedly Increased Particle Emissions from the Steel Industry Determined by Wet/Semidry/Dry Flue Gas Desulfurization Technologies (Li et al, Environ. Sci. Technol. 2019, 53, 17, 10361-10370)
Although extreme weather is likely to overtake it in the near future, the deadliest component of dangerous fossil fuel waste has been air pollution, which kills 7 million people per year, a portion of the death toll resulting not from the combustion of dangerous fossil fuels, although they dominate air pollution, but from the combustion of biomass. The chief component of air pollution that kills people is particulate matter, although the acid gases (sulfur oxides and nitrogen oxides) and ozone also contribute on a fairly grand scale.
There are many technologies for addressing sulfur oxides and I've written about some here recently. Not all dangerous fossil fuels are consumed for power plants and transport devices; some are consumed for material usage. The paper under current discussion suggests that there is no free lunch, using one technology can impact others.
From the introduction:
Pollutant emissions from the nonpower industry have recently attracted increasing attention as CFPPs have significantly reduced their emissions, especially those from one of the major industrial sources, i.e., steel plants.(11?13) A total of 17.1–36.9% of the atmospheric PM2.5 in many industrial cities has been attributed to emissions from steel plants, as suggested by source apportionment investigations.(14?16) China has been the largest steel producer in the world since 1996 (837.7 Mt in 2017, approximately 49.2% of the total production in the world).(17,18)Figure S1 shows that the relative contribution of primary PM2.5 emissions from the steel industry to total anthropogenic emissions grew from 5.4 to 8.2% from 2005 to 2014 in mainland China, while the relative contribution from CFPPs decreased from 9.5 to 5.1% in the same period, mainly attributed to the ultralow-emission requirement. Because steel emission standards lagged behind those for CFPPs during this period, the steel industry has emitted more PM2.5 than CFPPs since 2008.(17,18) Aiming to improve local air quality, Hebei Province in North China has implemented an ultralow-emission standard for steel plants starting in 2019.(19) The emission parameters for sinter flue gas in the Hebei ultralow-emission standard are the same as those for CFPPs. Although there is still no national standard for steel plants similar to the ultralow-emission standard for CFPPs deployed in mainland China, the detailed requirements of pollutant emissions for the steel industry have recently been under discussion with regard to standard feasibility and flue gas complexity.
The whole iron and steel producing process, mainly including sintering/pelletizing facilities, blast furnaces, basic oxygen furnaces, electric arc furnaces, and steelmaking furnaces, can generate pollutant emissions.(20,21) The sintering process is the major emission source of most pollutants, including PM, SO2, and NOx, with relative contributions of approximately 30–45, 70, and 90%, respectively, in the whole process.(13,22) Sintering flue gases are generally much more complex than those of CFPPs, exhibiting traits such as variable concentrations/compositions of pollutants, including SO2 and unknown corrosive gases, large temperature fluctuations ranging from 80 to 180 °C, and high variations in oxygen content ranging from approximately 10 to 15%...
It appears that desulfurization technologies designed to address acid gases have had an effect on the particulate emissions.
The wet flue gas desulfurization discussed herein involves the use of limestone and ammonia.
Some pictures from the text:

The caption:

The caption:

The caption:

The caption:
The caption:

The caption:
It must be said however, that the authors state that it is not clear that the particulates involved desulfurization technology while contributing to Chinese haze, are not the normal carcinogens associated with coking.
Some text:
Figure 5 summarizes the relative contributions of the newly generated components from the FGD systems to PM emissions in the stack. The estimation of newly generated components originating from FGD systems is estimated based on the assumption of the most abundant metal elements (i.e., K and Fe), which are increased by FGD desulfurizers and their byproducts (see the description in the Supporting Information text). Relative mass ratios of 16.5, 63.4, 59.4, and 70.7% in the emitted PM2.5 components are replaced by FGD desulfurizers and their byproducts for limestone WFGD, ammonia WFGD, semidry CFB-GFD, and activated coke dry FGD systems, respectively. The 16.5% replaced components of PM2.5 in the limestone WFGD outlet are occupied by SO42– (9.1%) and Ca (2.3%), while the 63.4% in the ammonia WFGD outlet are dominated by SO42– (43.1%) and NH4+ (14.2%). The 59.4% increase in components in the CFB semidry FGD outlet was mainly attributed to Ca (22.6%) and SO42– (13.1%), and the higher proportions of other compounds may be derived from impurities of the desulfurizer itself or unreacted hydrated lime. The PM2.5 in the activated coke dry FGD outlet possesses a high ratio of EC (39.9%), OC (14.4%), and NH4+ (14.2%). The proportion of byproducts in PM2.5 in the ammonia WFGD, activated coke dry FGD and CFB semidry FGD is over 60%, but this is not the case in the limestone WFGD. The smaller effect on PM2.5 component characteristics at the limestone WFGD outlet could be attributed to its relatively mature desulfurization process (optimal size of spraying slurry droplets) and the efficiency of removing entrained droplets of enriched byproducts by its demister and WESP. Limestone WFGD is the dominated technology (about 90%) for reducing SO2 in CFPPs. Since the concentration of PM is greatly influenced by the scouring intensity of desulfurization slurry and the effect of flue gas carrying. High-efficiency WESP has been commonly installed in front of the stack to effectively reduce the final PM emission to meet the “ultralow-emission standard” in CFPPs recently.(5) Compared to that of CFPPs, more attention about the newly generated components from the FGD systems to PM emissions should be paid for the steel industry.
Interesting.
In any case, the steel industry is an environmental problem that is not generally addressed in some of the wishful thinking we hear about climate change.
Have a pleasant Sunday evening.
Profile Information
Gender: MaleCurrent location: New Jersey
Member since: 2002
Number of posts: 33,512