Environment & Energy
Related: About this forumAn interesting reactor H2O free approach for the direct synthesis of the wonder fuel DME.
Regrettably I won't have much time to fully discuss this paper: Kinetics, Mass Transfer, and Reactor Scaling Up in Production of Direct Dimethyl Ether, Kainakhone Pathoumthong, Putong Ratanamalaya, Sunun Limtrakul, Terdthai Vatanatham, and Palghat A. Ramachandran Industrial & Engineering Chemistry Research 2022 61 (46), 17077-17091.
I like it because it has a very nice summary of why DME is the perfect energy currency for the localized distribution of the energy of nuclear heat, since it is a drop in substitute, more or less, for the infrastructure built for the use of dangerous fossil fuels. In short it can replace diesel fuel, gasoline, LPG, methane in all applications without the use of extreme conditions, since its critical temperature is around 150°C, meaning it is easily liquified without the need for refrigeration.
From the text:
The authors discuss two reactor approaches commonly used in DME synthesis:
They then note that a problem in direct DME synthesis from syn gas (H2 + CO or H2 + CO2) is that the reaction is highly exothermic, it generates significant heat. The heat can lead to the deactivation of the catalyst, and thus an efficient means of temperature control is required. They then discuss and evaluate a biphasic bubbling reactor, where a liquid phase (typically molten wax) is used to control temperatures, presumably allowing for the recovery and use of the heat in process intensification settings.
The authors discuss issues in scaling these types of reactors to a required industrial scale.
The reactor schematic looks like this:
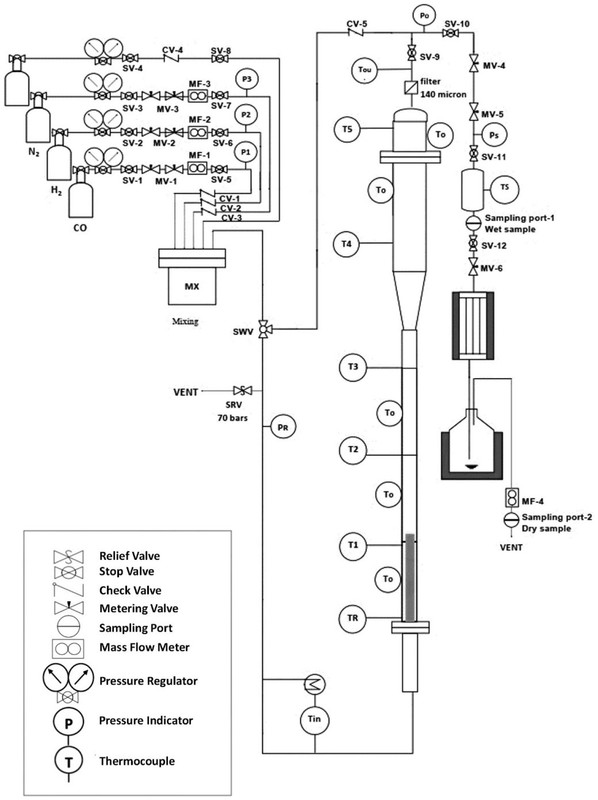
The caption:
What I find interesting, an issue that sometimes troubles me when I think about DME systems is that the water generated is consumed directly, avoiding the somewhat more difficult separation of DME and water for the easier separation of CO2 and DME, whereupon the CO2 can be recycled (perhaps by dry reforming of waste materials) back to CO for reuse.
The reactions in the continuous one step system are given as follows:

Again, I won't have time to go into more detail on this interesting and exciting paper, but this kind of chemistry can save the world, provided the primary energy source is nuclear.
Have a nice day tomorrow.
Backseat Driver
(4,671 posts)controls of end user diesel vehicles? If not, working out the kinks in production as well as OSHA stuff for worker safety--seems sort of do-able, and by-products of end-users burning this fuel seem both non-toxic with less wear-and-tear on engine parts while by-products of production could be re-cycled for other uses? Seems a good reason for keeping "nuclear" options open, but I'm totally clueless about industrial chemistry.
NNadir
(38,532 posts)Some seals used in diesel engines powered by dangerous fossil fuels are not compatible with DME, as I understand it. DME has lower lubricity than diesel fuel (not surprising) and thus small amounts of lubricant may be required as an additive.
We already have diesel trucks that run on methane, chiefly those associated with waste companies, since landfills often generate methane. (I favor replacing landfills with dry reforming or steam reforming systems driven by nuclear heat. The resulting syn gas might be processed into any of the things now provided by petroleum or into DME.) DME can be used anywhere methane is used, except DME, unlike methane is not a greenhouse gas; it's atmospheric lifetime is about 5 days.
The energy/volume density of DME, although its cetane number is higher than petroleum diesel fuel, is lower. Thus the fuel tank would need to be slightly larger or alternatively, the driving range lower. The storage of the fuel is almost exactly the same as that of LPG, so existing LPG infrastructure would be able to drop in directly to DME.
All existing pipelines can be easily switched to carry DME, preferably in a liquified form.
It is an excellent refrigerant by the way, and it is also a fairly decent thermal fluid, it can be utilized to transfer heat via a system that might allow as a long distance heat pump.
It really is a wonder fuel. It sort of amazes me that the value has not been generally recognized. Mostly today it's used as a propellant in hair spray, having replaced CFCs. It's kind of crazy.

Kick in to the DU tip jar?
This week we're running a special pop-up mini fund drive. From Monday through Friday we're going ad-free for all registered members, and we're asking you to kick in to the DU tip jar to support the site and keep us financially healthy.
As a bonus, making a contribution will allow you to leave kudos for another DU member, and at the end of the week we'll recognize the DUers who you think make this community great.