Environment & Energy
Related: About this forumRecent Advances in Cladding Material Extraction from Fuels in Nuclear Fuel Cycles
The paper to which I'll refer in this post is this one: Recent Advances in Cladding Material Extraction from Fuels in Nuclear Fuel Cycles Kunal Mondal and Richard T. Mayes Industrial & Engineering Chemistry Research 2026 65 (4), 1915-1928.
The authors are out of Oak Ridge National Lab, still one of the premier nuclear laboratories in the world, at least until our government of ignoramuses fucks it up.
I am very glad to see this paper, since the matter of zirconium has been on my mind for some time. This is a review article, and thus rather long, and I can thus, only scratch the surface and perhaps reproduce a few figures.
The amount of used nuclear fuel in the United States, despite much angst and carrying on by people who know next to zero about the subject, is small, as industrial products go, somewhere between 80,000 and 90,000 metric tons, vanishingly small when one considers the scale of automotive waste, fossil fuel waste - especially highly toxic coal ash and petroleum by products, never mind the trillion ton quantities of the fossil fuel waste carbon dioxide that have been dumped over the years - aluminum red mud, electronic waste - including solar cells from the useless PV industry - and even turbine blade waste from the useless wind industry. Moreover, used nuclear fuel is largely ceramic in nature and is chemically, for the most part, easy to contain since most components, are insoluble in water, famous exceptions like 137Cs (a wonderful material in my view) notwithstanding.
Actually the nature of used nuclear fuel is an advantage for nuclear technology, as opposed to the fearsome assignment in the public imagination as a subject of terror. There is little accounting for how the public can be whipped up to mass paroxysms stupidity, is there?
Let me reproduce some text from the paper before discussing zirconium in any detail. From the introduction:
Fission product recovery has been carried out as part of ongoing SNF research. (6) Recovery of cladding materials from SNF has drawn wide interest for several reasons, (7,8) including its potential to minimize radioactive waste disposal. (9) Cladding materials become contaminated by nuclear fuel, so appropriate nuclear waste management is required. (10) Recovery and recycling of cladding material can provide a sustainable solution to address environmental concerns by minimizing the volume of wastes that must be stored for long periods ranging up to thousands of years. Recovery and recycling also can minimize the risks associated with nuclear waste disposal. (11)
The strong economic motivation for recovering cladding materials (12,13) is based on their limited availability and the crucial need for them in the nuclear industry. (14,15) Recovery processes of these strategic resources enable reclamation of valuables, therefore reducing the demand on primary production and providing a more economic, more sustainable supply chain. (16,17) Recovery and reuse of these materials contribute to a circular economy approach in which resources are recovered and dependence on raw material extraction is reduced. (18)
Beyond waste-management advantages, the value of zirconium alloys themselves reinforces the motivation for recovery. (19) Although zirconium is not a rare earth element and its minerals are relatively abundant, producing nuclear-grade zirconium demands multiple refining steps, including Zr–Hf separation, and depends on a small number of qualified suppliers. (20) These factors elevate the cost of zirconium metal and nuclear-grade alloys to tens of US dollars per kilogram... (21)
I added the bold to point to major issues with zirconium, the chief component, as we shall see, of the cladding of accumulated used nuclear fuels . Let me elaborate.
Because of an effect known as the "lanthanide contraction" between the 4th and 5th periods of the periodic table, resulting from the extension (and reduced shielding) of the f orbitals electrons added to the lanthanides from lanthanum to lutetium, the elements in groups 3, 4, 5, and 6 in those periods, respectively the yttrium/lanthanum (and other lanthanides), zirconium and hafnium, niobium and tantalum, molybdenum and tungsten, are always found in ores with one another and, in addition, are very difficult to separate from one another. This last effect relates to their similar atomic radii.
The same conditions would have applied for the separation of technetium from the rare, valuable, and industrially important element rhenium, if technetium occurred on Earth, which it doesn’t in visible amounts. Technetium, element 43, is the lightest element in the periodic table for which no stable isotopes exist, all of its isotopes are radioactive, and only one isotope, 99Tc, exists in sufficient quantities to be put to use. All of the technetium that can be isolated at a ton scale are present in used nuclear fuel. Regrettably, in some existing industrial (Purex) separation schemes, notably those at La Hague and (once upon a time) those at Sellafield, technetium was dumped in the ocean as the very soluble TcO7- ion.
As far back as the 1960’s this situation, dumping technetium, was considered appalling by some workers experienced with the element. (cf. A. K. Lavrukhina, A.A. Pozdnyakov, Translated by R. Kondor, Israel Program for Scientific Publications, Analytical Chemistry of the Elements, Technetium, Promethium, Astatine, Francium, from the Series prepared by the Vernadskii Institute of Geochemisty and Analytical Chemistry, USSR, Ann Arbor-Humphrey Science Publishers, (1970) Page 8.) This had nothing to do, despite what popular fancy may emote to thinking otherwise, to do with the fact that technetium is radioactive: Since it’s chemistry, and, except to a minor extent, it so similar to valuable rhenium, this suggests that despite its radioactivity, its similarity calls forth many applications, since it is relatively straight forward for the supply of technetium to exceed that of rhenium. This is particularly true of alloys dependent on rhenium, particularly some “superalloys.” The concentration of technetium in Purex raffinates is small, according to the 1960’s reference just cited, between 5 and 50 mg/liter, and probably would be somewhat, but not hugely, higher now, given the greatly increased burnups of modern nuclear fuel. It is also notable than almost all of the nuclear fuel reprocessing in that time was connected with the manufacture of nuclear weapons, not commercial nuclear fuel.
Recently in this space, in preparation of a post about technetium, I posted about the concept of a “Sherwood Plot,” a concept developed in the 1950’s, unsurprisingly, by Thomas Sherwood, which linked the sustainable costs of a system with its concentration, and with the cost. Commercial fuels are extremely likely to contain much higher concentrations of technetium in its PUREX raffinates, than weapons processed fuels.
The reason is this: Nuclear fuel used in reactors designed for weapons production is very different than commercial fuel, because of the physics of plutonium isotopes suitable for use in weapons, essentially limited to being as pure 239Pu as is achievable with acceptable mass yields. Thus the fuel must be irradiated with neutrons for a vastly shorter period than is desirable for commercial fuels to avoid the production of 240Pu, higher isotopes and the lower isotope 238Pu. (Weapons grade plutonium is far more expensive and has greater environmental impacts than reactor grade plutonium.) By contrast, it is desirable to keep commercial fuels irradiated for as long as possible, and the opposite is true for the preparation of plutonium from them. the alternate isotopes, in particular 238Pu but also 240Pu are desirable to prevent diversion of plutonium to weapons purposes. Under the right conditions, plutonium, the synthetic element with the potential to save the world, can be rendered essentially useless for the production of nuclear weapons by the "denaturing" of pure or nearly pure 239Pu with 238Pu, 240Pu, and 242Pu, while still retaining the fissile value of 239Pu and in situ generated 241Pu. Of course, longer irradiation means more fission products and thus higher technetium concentrations is separation residues, whether from PUREX, TRUEX, GANEX or other conceivable extraction processes, or in processes that are likely to be superior to extraction processes.
Which returns me to the point of zirconium, the chief constituents of zircalloy, the alloy currently utilized in most nuclear fuels, with rare exceptions. The paper gives the elemental composition of several commercial grades of zircalloy:
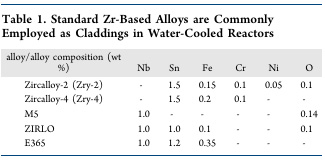
These distributions of elements are not arbitrary; they involve considerable development and the components are each there for a reason. Niobium, as a source material, has interesting implications in relationship to zirconium. Niobium, which as is described above is always found with tantalum in ores is monoisotopic: It contains only one stable isotope, 93Nb, which interestingly, although the mass number 93 is one of the most prominent mass numbers among the fission products of 235U as well (to a slightly less extent) 239Pu, and yet it is really not possible to isolate pure non-radioactive niobium from among fission products. The reason for this is that the radioactive precursor to 93Nb is 93Zr, an isotope of zirconium which is one of the seven long lived fission products generated by nuclear fission.
These are 79Se, (selenium) (t1/2 = 327,000 years,) 93Zr (zirconium) (t1/2 = 1,600,000 years), 99Tc (technetium) (t1/2 = 211,100 years), 107Pd (palladium) (t1/2 = 6,100,000 years), 126Sn, (tin) (t1/2 = 230,000 years), 129I, (iodine) (t1/2 = 15,900,000 years) and 135Cs (cesium) (t1/2 = 1,300,000 years).
The long half-life of 93Zr means one would need to wait tens of thousands of years to obtain even small significant amounts of pure nonradioactive niobium from used nuclear fuel, and then only under conditions in which the zirconium were separated from the other components. The paper cited
I recently discussed the use of 107Pd (palladium), which is formed along with nonradioactive isotopes of palladium, and, earlier, the very minor fission product 79Se, (selenium). The paper cited at the outset is all about 93Zr.
The zirconium in used nuclear fuel comes from two sources, mining, where it needs to be separated, as noted in the text from its congener, hafnium, which is materially very similar to zirconium in its mechanical properties, but very different in its nuclear properties and as a fission product, where all of its natural isotopes are formed as well as the long lived radioactive isotope 93Zr .
Hafnium's isotopes have large neutron capture cross sections, which would cause a reactor to shut down, which is why, it is sometimes used, generally in small naval reactors, in control rods. As noted, this process of separation is expensive. (The industrial scale separation of hafnium and zirconium on an industrial scale was one of the unsung great scientific achievements of the Manhattan Project.)
It is possible to obtain pure nonradioactive monoisotopic zirconium, 90Zr from used nuclear fuel by separating, before it decays with its 28 year half life, the famous radioactive isotope 90Sr, strontium 90, a pure beta emitter (as is the intermediate 90Y, which has a half-life of 64 hours). 90Sr has been used as a portable power source in RTGs, and could still be so used in a situation where nuclear fuels are routinely reprocessed. In theory, particularly in a sustainable world where all, or almost all primary energy was nuclear, small electrical energy systems could be, (and actually were in the former Soviet Union) used in remote places to provide continuous power by similar technology as that which powers the most remote objects ever made by human beings, the Voyager space craft. Unlike Voyager, 90Zr, might be harvested periodically. (As it happens, 90Zr has superior nuclear properties, neutron transparency, than all of the other naturally occurring zirconium isotopes, 91Zr, 92Zr, 94Zr, and 96Zr. 96Zr, which makes up around 2% of natural zirconium, is known to be radioactive, albeit with a half-life much longer than the age of the universe, and 94Zr, is suspected, but not proved, to be radioactive as well. If it is, its half-life will be longer than that of 96Zr and its radioactivity negligible.
Small amounts of radioactive 93Zr, will be formed by neutron capture in 92Zr in natural mined zirconium, but similarly 93Zr will be transformed into 94Zr by the same process, with an equilibrium being established by model solutions of the coupled Bateman equations for mass numbers between 88 and 96:

In any case, zirconium, although this is often ignored, is the second largest component of bulk used nuclear fuel after unreacted uranium as shown in this figure from the paper:
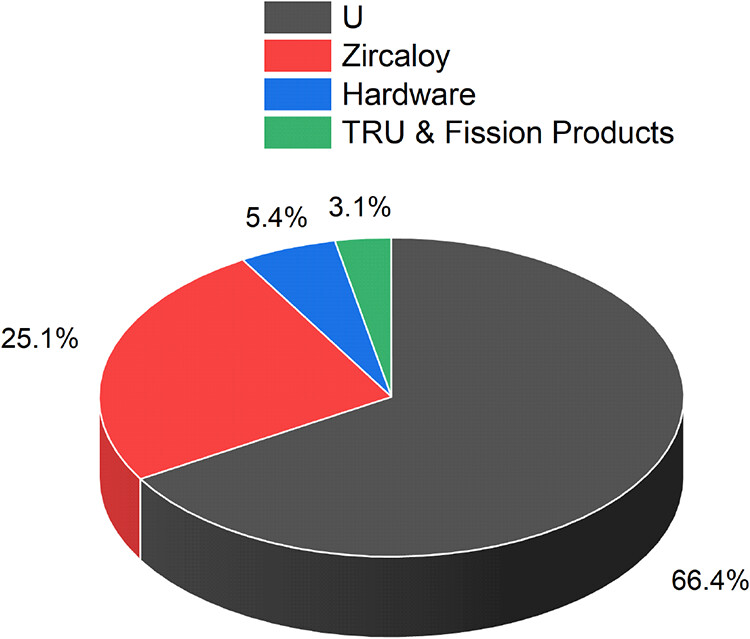
The caption:
The high energy to mass density of actinide nuclear fuels that makes nuclear energy vastly superior in an environmental sense to all other forms of energy means that even under current circumstances, where uranium is still mined from ores (although it need not be so for the long term), that nuclear energy minimizes, but does not eliminate, the need for tearing the shit out of the planet's rocks to isolate metals and relatively rare semiconductors. This is why it is vastly superior to all of the so called "renewable energy" junk that our antinukes hype while not giving a shit about fossil fuels. However relatively small amounts of metals are necessary for modern nuclear reactors, zirconium being such an element. However, nuclear energy has the otherwise unknown property in any other form of energy inasmuch as its operations actually increase the amounts of materials needed for its operations, as is well known by the thorium/uranium couple, the uranium/plutonium couple, as well as that of the minor actinides. Zirconium as a fission product is yet another example, were the suggestion of the paper embraced.
When I wrote about the recovery of palladium from used nuclear fuel in this space recently, (Improved efficiency of selective photoionization of palladium isotopes via autoionizing Rydberg states) I was able to suggest what the estimated concentration of radioactive 107Pd among the palladium isotopes would be, around 17%. This would not be possible, estimating the dilution factor of the single long lived radioisotope in the case of zirconium, since it will be a mixture of natural and fissiogenic zirconium. One can be fairly certain that it would be relatively low, and like 107Pd, 93Zr is a pure β emitter, with any penetrating radiation being connected with Bremsstrahlung effects, and likely of minor consequence. It is worthy of noting that in establishing a Bateman equilibrium, the neutron capture cross section of 93Zr is slightly higher than the other zirconium isotopes, and thus its destruction would be favored over its formation by neutron capture in 92Zr, which is more transparent to neutrons. Also the dilution factor would be a function of the cooling time provided to the used nuclear fuel from which zirconium is isolated, since 90Zr's formation is delayed by the half-life of 90Sr. Very old fuels, like say those available in my home state of New Jersey from the fuels in the early 1970s that powered the Oyster Creek Nuclear Reactor would be more diluted than fuels isolated from recently removed fuels from the magnificent Diablo Canyon Reactor in California.
However, from the pie chart, figure 3 from the paper above, it is clear that the overwhelming bulk of used nuclear fuels can be recovered for further use, particularly if we embrace, as we should, reactors with a fast neutron spectrum and in the case of thermal reactors, the addition of thorium to the fuels along with plutonium.
None of the above will prevent antinukes, given their poor understanding of nuclear science, as well as many other sciences, their absurd paranoia by which they make the morally dubious calculation that millions of deaths each year can take place from fossil fuel waste as long as no one on Earth ever dies from radiation exposure, and their indifference to the climate, health and ecological effects of fossil fuels and the vast destruction they are causing, from pushing their unsustainable so called "renewable energy" junk.
However, as bad as things are now, it strikes me as possible that a better world might exist, despite the dictatorship of ignorance that permeates our world culture as of modern times. "Possible" has a very different meaning that "likely" though, and while it is easy to be cynical, hope is not dead.
Have a nice evening.