Environment & Energy
In reply to the discussion: Elevated Radium Activity in a Hydrocarbon-Contaminated Aquifer [View all]NNadir
(38,261 posts)I think I already more or less answered this question, by showing the thermal efficiency of electrolysis to be roughly 85% at best, although for a real useful measure it matters when the solar energy is produced.
Thus, in a very, very, very (almost uselessly) crude way you would need 1/.85 = 1.18 EJ, not counting compression and liquefaction. If your storage tank was very large and very strong, you may avoid liquefaction, but with liquefaction, 1.18/0.7 = 1.68 EJ. In either case you would still need compression.
Again the embodied energy of the systems, pipes, tanks, compressors are ignored.
This will involve, at 120 MJ/kg LHV about 14 billion kg of hydrogen, and 8 times that mass of water ignoring evaporative losses, because we choose to be sloppy and as crude as possible. Since the molecular weight of hydrogen is 1/8 that of water, you would need, again ignoring evaporative losses from resistance heat, about 112 billion kg of water.
But again, this is uselessly crude, particularly because hysteresis and low current conditions are ignored.
1 EJ represents, as their are 86400 seconds in a day, an average continuous power of 11,600 MW.
Many times, particularly on cloudless days, solar output has a (mildly) Gaussian aspect. Near the most recent Equinox in California, (CAISO) that of this past March, there seems to have been clouds and or rain drifting in around this time, and the autumn equinox was also a little bit noisy, so I selected 9/24/2022, near the autumn equinox for the following graphics showing near the peak, the rough 1/2 height in the morning, in this case 8:00 am and the afternoon half-height, 19:25:
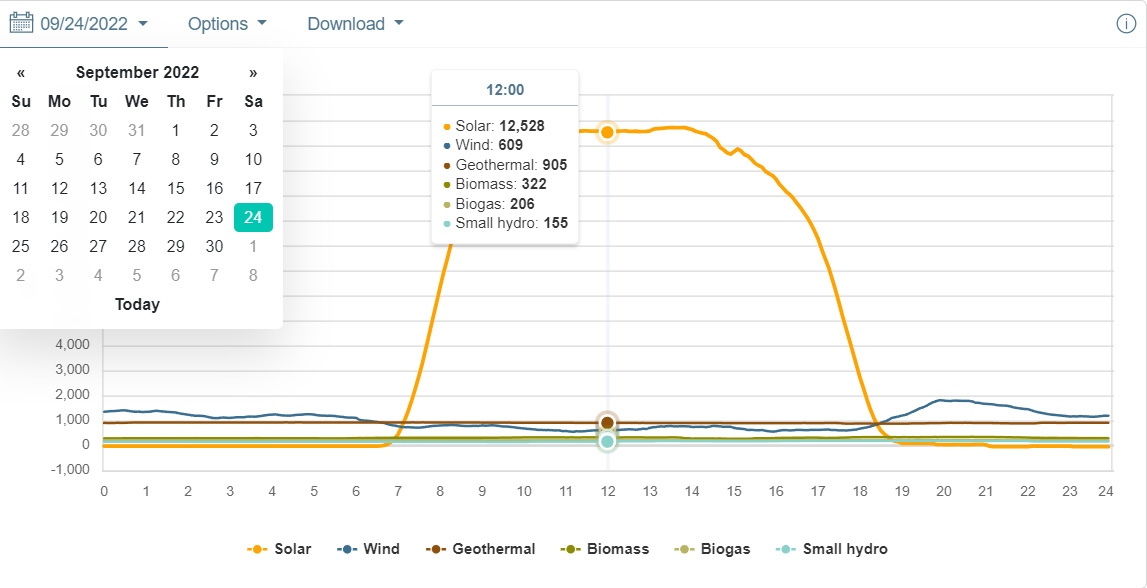
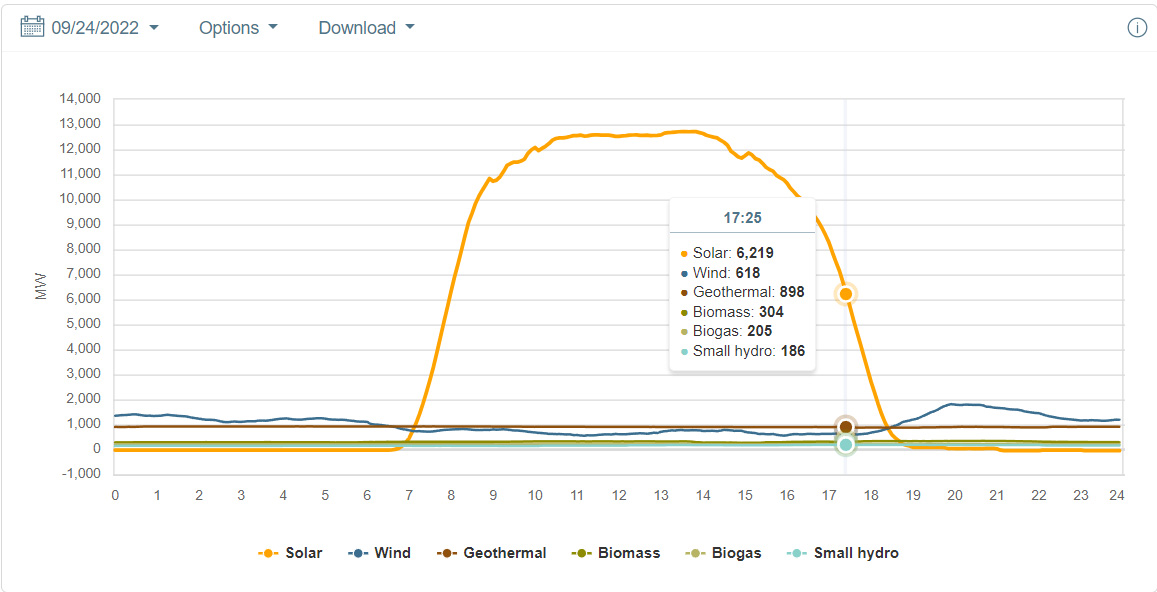
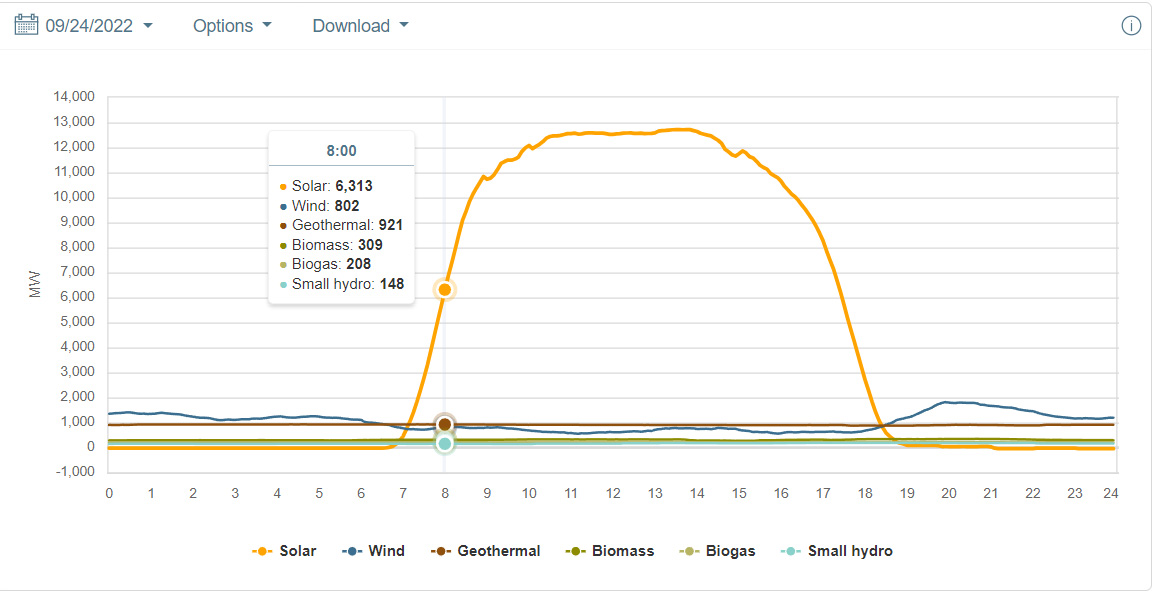
The peak was not quite at noon, but noon is close enough. This is important because the thermodynamic efficiency for the electrolyzers will change with the current.
At noon, you're storing energy, at half height you're breaking even, and you're burning hydrogen before half height in the morning, and after half height in the afternoon. You have about 9 and a half hours where you're storing energy as hydrogen, and 15 and a half hours where you're burning it to keep the power level constant. This will surely increase the 1.68 EJ to a higher number.
This will impact your tank size, and thus your external costs.
How you recover the energy will also have bearing. Presumably you would want to be efficient and use the power plant as a combined cycle device, with a thermal efficiency of around 60%, since fuel cells would need to be very large, get very hot, and would require an enormous amount of fluoropolymers.
At 60% thermal efficiency, achievable with a high end combined cycle plant, this brings you up to 1.68/0.6 = 2.80 EJ (liquefaction).
At the 40,000 sq km/EJ from my previous post, this brings the land area required (Mohave desert) to 40,000*2.8 = 112,000 sq km, about 1/4 the land area of California for 1 EJ produced in a single day at an average continuous power of 11,600 MW.
At the winter solstice a bigger tank would be required, as smaller one at the summer solstice but the "close to the equinox" value should be good for a first approximation, ignoring efficiency changes at high current and low current, embodied energy, etc, etc.
I hope this BOE calculation helps.