NNadir
NNadir's JournalFrom bomb to Moon: a Nobel laureate of principles
When I was a kid, I used to spend a lot of time at Urey Hall on the UCSD campus. Urey, of course, is the discoverer of deuterium.
At that time of my life I didn't think all that much about Urey, which was my loss. (I was a stupid kid and am now a somewhat less stupid adult.)
Nature has a review of a biography of Urey, and I'm going to put it on my list of "need to read someday."
From bomb to Moon: a Nobel laureate of principles
I think the review is open sourced, but if it isn't, some excerpts:
After witnessing the 1945 Trinity atomic-bomb test, the theoretical physicist J. Robert Oppenheimer recalled Hindu scripture: “Now I am become Death, the destroyer of worlds.” Although this is often interpreted as admitting moral culpability on the part of the Manhattan Project’s scientific director, Oppenheimer remained a central player in the nuclear-weapons establishment until he lost his security clearance in the mid-1950s.
Harold Urey also worked for the Manhattan Project. But by contrast, the Nobel-prizewinning chemist distanced himself from nuclear weapons development after the war. His search for science beyond defence work prompted a shift into studying the origins of life and lunar geology. Now, the absorbing biography The Life and Science of Harold C. Urey by science historian Matthew Shindell uses the researcher’s life to show how a conscientious chemist navigated the cold war.
Shindell argues that Urey’s pious upbringing underpinned his convictions about the dangers of a nuclear arms race, and his commitment to research integrity. Urey grew up a minister’s son in a poor Indiana farming family belonging to a plain-living Protestant sect, the Church of the Brethren. Progressing through increasingly diverse educational environments, culminating in a PhD at the University of California, Berkeley, Urey became self-conscious about the zealousness of his family’s faith. He also found the path to a cosmopolitan, middle-class life.
In the 1920s, Urey was among a small group of chemists who collaborated closely with physicists. Working at Niels Bohr’s Institute for Theoretical Physics at the University of Copenhagen, he kept abreast of developments in quantum mechanics. There, and on travels in Germany, he met the likes of Werner Heisenberg, Wolfgang Pauli and Albert Einstein. But Urey decided he lacked the mathematical skills to make theoretical advances in quantum chemistry. Moving back to the United States, he started both a family and an academic career.
At Johns Hopkins University in Baltimore, Maryland, and later at Columbia University in New York City, Urey taught quantum mechanics to chemists, while setting out on the trail that led him to deuterium. In 1931, he discovered this isotope of hydrogen. Predicted on the basis of work by Bohr, Frederick Soddy, and J. J. Thomson, its existence had been doubted by many chemists and physicists. Urey’s identification won him the Nobel three years later. By this time, he had also co-authored one of the first texts in English on quantum mechanics as applied to molecular systems, the 1930 Atoms, Quanta and Molecules.
Urey’s continuing work on stable isotopes of other chemical elements, such as nitrogen and oxygen, led to important applications in biochemistry and geochemistry, including the pioneering use of isotopic labels to study metabolic pathways. Living in New York also led Urey to political liberalism. He became aware of the anti-Semitism affecting Jewish scientists, and the lack of opportunities for women scientists. A generous mentor, he shared his Nobel prize money with two collaborators, and split a grant he had been awarded with the young Isidor Rabi (who later discovered nuclear magnetic resonance)...
...The Second World War changed Urey’s life, as it did those of most physical scientists and researchers in many countries. His expertise in isotopes made him valuable to the Manhattan Project. Here, he eventually headed a massive team of scientists and engineers working on the separation of uranium isotopes using gaseous diffusion methods. However, he was ill-suited to the pressure of managing this technologically complex and cumbersome project, and Leslie Groves — the project’s overall director — regarded him with suspicion. Even before the war’s end, Urey became deeply disenchanted with working for the military...
...After the war, Urey used his laureate status to voice alarm about the prospect of nuclear warfare. He backed international control through world government as a way to control the military future of atomic energy. This was not a radical view in 1946...
...Over this harrowing period, Urey lost faith in the ability of modern secular society to manage the new threats of the atomic age. Although he had long abandoned his parents’ religion, he began to argue that Judaeo-Christianity was key to democracy. He attributed the success of science itself, with its commitments to honesty and credit, to religious ethics...
...In the late 1940s, Urey used his expertise in mass spectrometry to begin work in geochemistry, and then in planetary science. It was a way to escape the orbit of the nuclear weapons establishment (although he still advised the US Atomic Energy Commission). With chemistry graduate Stanley Miller, he tested hypotheses on the origins of life by Soviet biochemist Alexander Oparin and biologist J. B. S. Haldane, and successfully produced amino acids by sparking a solution of water, methane, ammonia and hydrogen. In 1952, Urey published The Planets, a chemical treatise on the formation of the Solar System...
...Urey became influential during the early days of NASA, formed after the 1957 launch of the Soviet satellite Sputnik, offering the agency persuasive reasons to prioritize exploration of the Moon over other bodies. In 1969, he analysed lunar rocks collected during the Apollo 11 mission, which supported his theory of the Moon’s common origin with Earth. He wanted the well-funded agency to test theories about the origins of the Solar System — experimentation beyond the reach of individual university scientists. Despite his influence, he was disappointed in this: NASA focused on crewed space exploration over questions of cosmogony.
Sounds like a cool book about a cool life, no?
Have a nice weekend.
The terrible day of the wisecrack.
From the Wikipedia entry on the life of Dorothy Parker:
These were no giants. Think who was writing in those days—Lardner, Fitzgerald, Faulkner and Hemingway. Those were the real giants. The Round Table was just a lot of people telling jokes and telling each other how good they were. Just a bunch of loudmouths showing off, saving their gags for days, waiting for a chance to spring them... There was no truth in anything they said. It was the terrible day of the wisecrack, so there didn't have to be any truth...[61]
Dorothy Parker
Of course, things are much worse in the age of Twitter, the age of anti-thinking.
We are all in the Algonquin Round Table, and that is not a good thing.
Mapping 123 million neonatal, infant and child deaths between 2000 and 2017
The paper I'll discuss in this post is this one: Mapping 123 million neonatal, infant and child deaths between 2000 and 2017.
This paper is open sourced, and anyone who cares can read it in its entirety.
The argument is often made - and it's a very good one - that the carrying capacity of the planet for human beings has been exceeded now for many decades. Thus it might seem that an argument for saving the lives of children under the age of five while consistent with human ethics may conflict with environmental ethics.
I have long argued that this conflict is actually invalid. The countries with the lowest birth rates are precisely those where people are secure in their homes, have sufficient health care, shelter, food, and where the rights of women in particular are most actively supported. The problem of exploding populations is therefore, in my opinion, is actually an issue of poverty and human development.
This is precisely why I personally focus the overwhelming portion of my private scientific interests on clean energy, because without clean energy, we cannot eliminate poverty and advance human development.
From article 25 of the Universal Declaration of Human Rights, approved by the United Nations in 1948, but since honored more in breach than practice:
(1) Everyone has the right to a standard of living adequate for the health and well-being of himself and of his family, including food, clothing, housing and medical care and necessary social services, and the right to security in the event of unemployment, sickness, disability, widowhood, old age or other lack of livelihood in circumstances beyond his control.
Universal Declaration of Human Rights
Note that Article 25 is not about electric cars and McMansions with solar cells on the roofs. Modern liberalism differs from 1948 liberalism; I personally prefer the latter.
From the abstract:
From the introduction:
Progress in child survival also diverges across age groups4. Global reductions in mortality rates of children under 5—that is, the under-5 mortality rate (U5MR)—among post-neonatal age groups are greater than those for mortality of neonates (0–28 days)4,8. It is relatively unclear how these age patterns are shifting at a more local scale, posing challenges to ensuring child survival. To pursue the ambitious Sustainable Development Goal (SDG) of the United Nations9 to “end preventable deaths of newborns and children under 5” by 2030, it is vital for decision-makers at all levels to better understand where, and at what ages, child survival remains most tenuous.
A map:
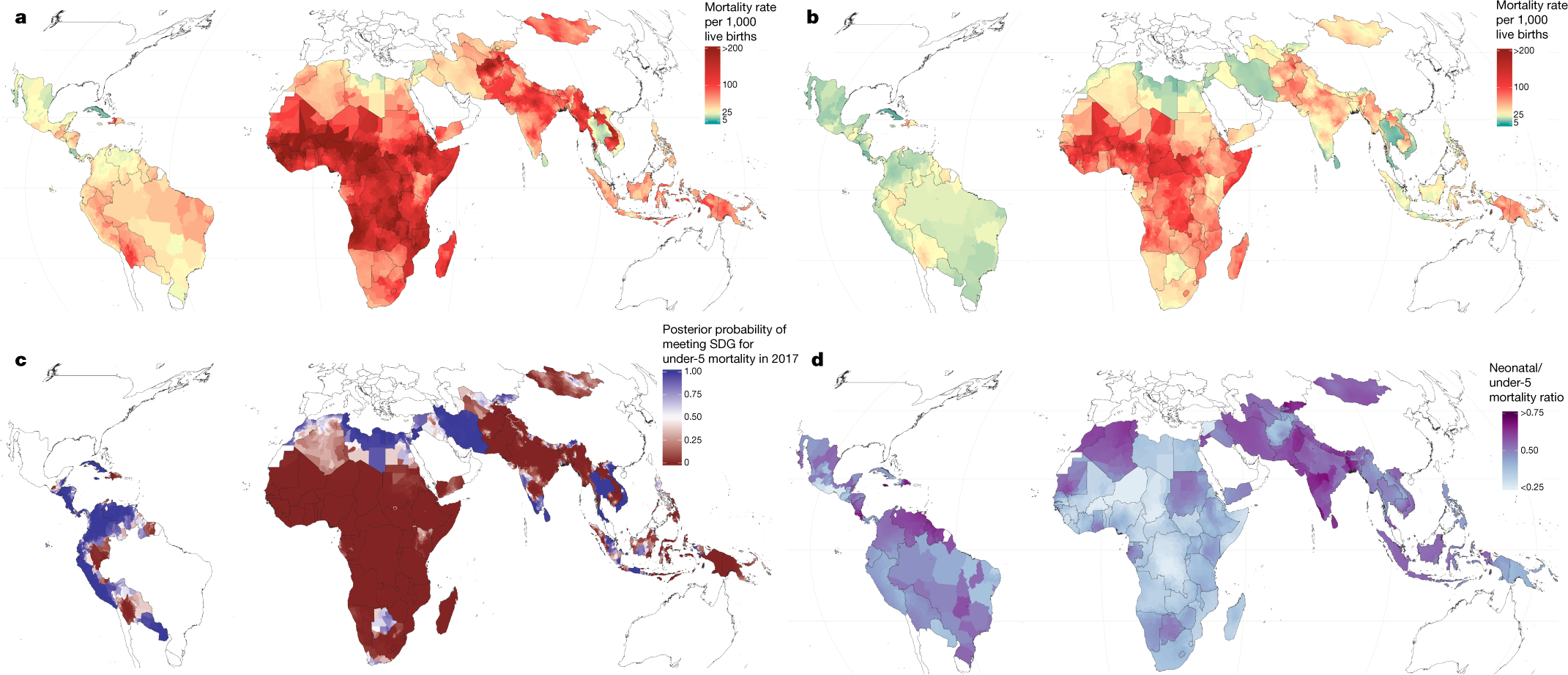
The caption:
We live in a country where children are kept in cages for no "crime" other than their race. We are beneath contempt.
This is probably one of the most important scientific papers in terms of ethical import I've read in a long time, and I read a lot of papers.
Have a nice weekend.
Crosslinking ionic oligomers as conformable precursors to calcium carbonate
The paper I'll discuss in this post is this one: Crosslinking ionic oligomers as conformable precursors to calcium carbonate (Tang et al, Nature 574, 394–398 (2019))
The fastest growing contributor on this planet in the 21st century has been dangerous coal, followed by petroleum, which is likely to be exceeded in the next decade by dangerous natural gas. The next largest contributor, also accelerating, is land use changes. Following these, closely is concrete. (Much of the climate change cost of concrete is connected to heat, almost always generated by the use of dangerous fossil fuels. In theory, if not in wide practice it is possible for the use of concrete to by carbon negative, and some major advances along this line have been made, for instance Riman Concrete, but even Riman concrete requires heat to make. Nuclear heat is actually the only practical way to make concrete without dangerous fossil fuels, despite whatever cartoons you've read or even written about solar thermal plants. Solar thermal plants didn't work, they aren't working and they won't work to address climate change and they will never work to make concrete.
In the past several years, my vicarious interest in my son's education has led me to consider a concept called "polymer derived ceramics" in which is just what it sounds like, a polymer is, via process engineering (and generally heat) converted into a highly structured ceramic. This paper touches on that concept, at least in a loose way. Beautiful things, these, with all sorts of fabulous potential applications.
The abstract, which is open sourced:
An excerpt from the introduction:
Published: 16 October 2019
Crosslinking ionic oligomers as conformable precursors to calcium carbonate
Zhaoming Liu, Changyu Shao, Biao Jin, Zhisen Zhang, Yueqi Zhao, Xurong Xu & Ruikang Tang
Nature volume 574, pages394–398 (2019) | Download Citation
Article metrics
556 Accesses
13 Altmetric
Metricsdetails
Abstract
Inorganic materials have essential roles in society, including in building construction, optical devices, mechanical engineering and as biomaterials1,2,3,4. However, the manufacture of inorganic materials is limited by classical crystallization5, which often produces powders rather than monoliths with continuous structures. Several precursors that enable non-classical crystallization—such as pre-nucleation clusters6,7,8, dense liquid droplets9,10, polymer-induced liquid precursor phases11,12,13 and nanoparticles14—have been proposed to improve the construction of inorganic materials, but the large-scale application of these precursors in monolith preparations is limited by availability and by practical considerations. Inspired by the processability of polymeric materials that can be manufactured by crosslinking monomers or oligomers15, here we demonstrate the construction of continuously structured inorganic materials by crosslinking ionic oligomers. Using calcium carbonate as a model, we obtain a large quantity of its oligomers (CaCO3)n with controllable molecular weights, in which triethylamine acts as a capping agent to stabilize the oligomers. The removal of triethylamine initiates crosslinking of the (CaCO3)n oligomers, and thus the rapid construction of pure monolithic calcium carbonate and even single crystals with a continuous internal structure. The fluid-like behaviour of the oligomer precursor enables it to be readily processed or moulded into shapes, even for materials with structural complexity and variable morphologies. The material construction strategy that we introduce here arises from a fusion of classic inorganic and polymer chemistry, and uses the same cross-linking process for the manufacture the materials.
Main
Many materials are consolidated from their crystallized powders16, but their resulting discontinuous internal structures render them brittle with a poor ability to resist fracture17,18. By contrast, polymeric materials are ubiquitous in modern society, due not only to their varied properties but also to their ease of fabrication15,19. The polymerization strategy is superior to crystallization because of its efficiency and controllability. In polymer chemistry, covalent bonds have an important role in ensuring the linkage of small units. Although a few covalent-bond-based inorganic materials (for example silicone and silica)20,21 can be obtained as polymers, there is no general method for the preparation of such materials by crosslinking owing to the lack of investigation into ionic monomers or oligomers for this purpose. In the control of polymerization reactions, a capping agent is key22: capping can stabilize precursors, whereas de-capping can initiate polymerization. Analogously, we proposed that ionic oligomers could be stabilized by an appropriate capping agent. Capping based on hydrogen bonding was thought to be suitable, because most inorganic complexes contain oxygen. For example, triethylamine (TEA) can form a hydrogen bond with a protonated carbonate through its tertiary amine group. More importantly, TEA is a small molecule that can be volatilized at room temperature, and it was expected that this could initiate an expected crosslinking reaction.
The authors use triethylamine in a solvent, the solvent in this case being ethanol (which of course, unless the ethanol is recovered, makes industrialization questionable. The calcium carbonate is made by bubbling carbon dioxide through an ethanolic solution of calcium chloride. Mass spectrometry demonstrated the existence of calcium carbonate polymers from trimers to undecamers, with, for some reason, nonamers excluded. The structures were also studied by 13C NMR.
A figure from the paper:
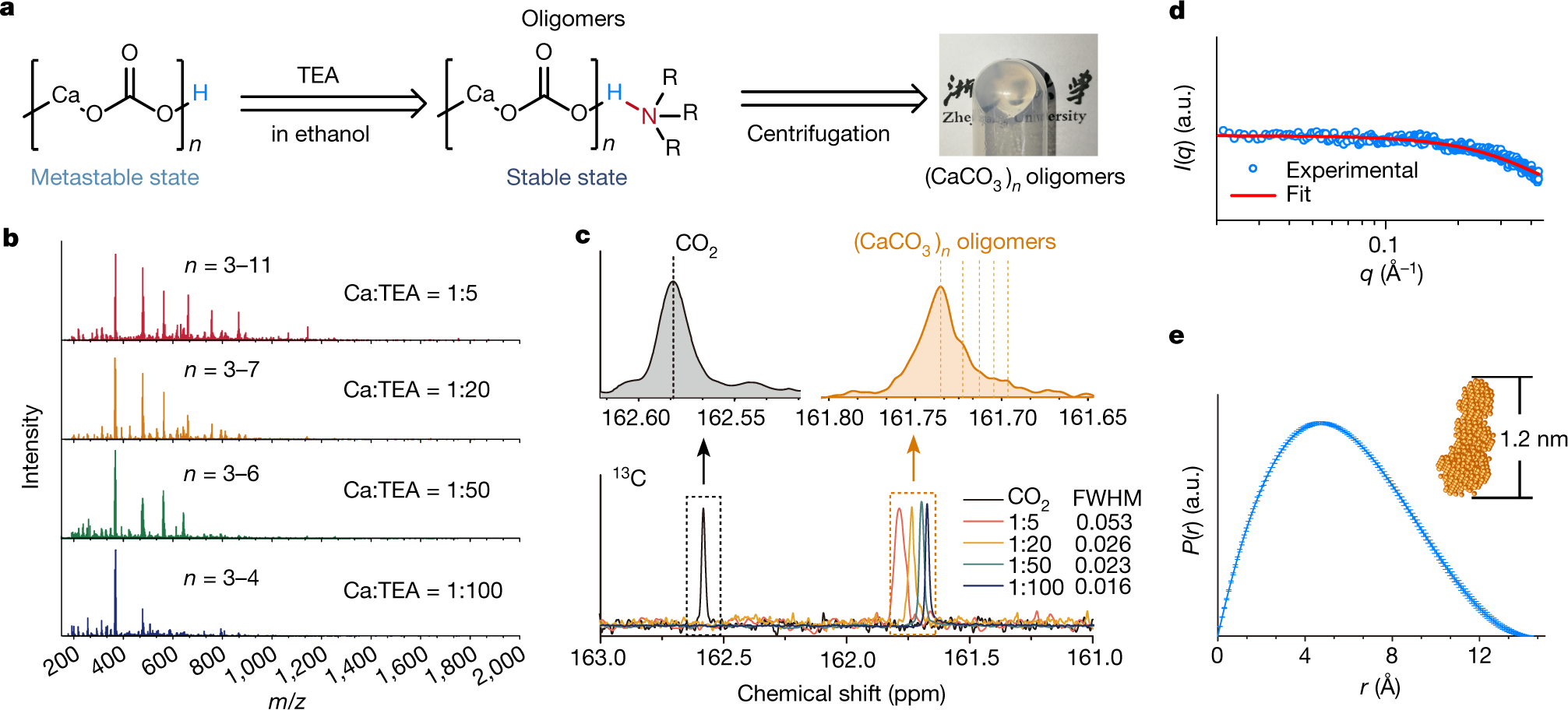
The caption:
The fate of the chloride ions is not reported. This seems to me to be an important consideration, nevertheless this is an interesting paper.
Cross linking of the polymers is achieved by evaporating the ethanol and the triethylamine.
Another graphic:
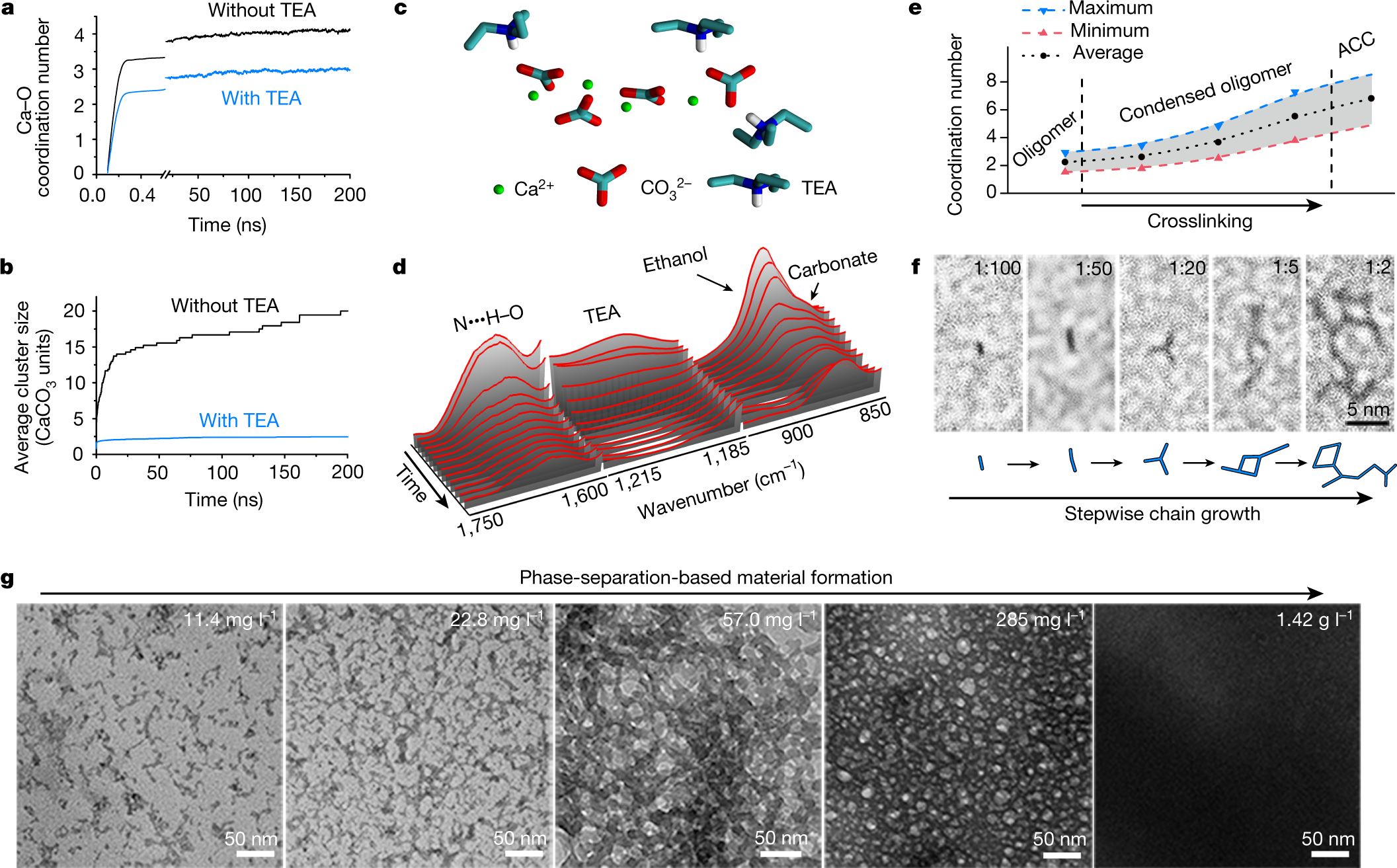
The caption:
Some text about the results:
Another picture:
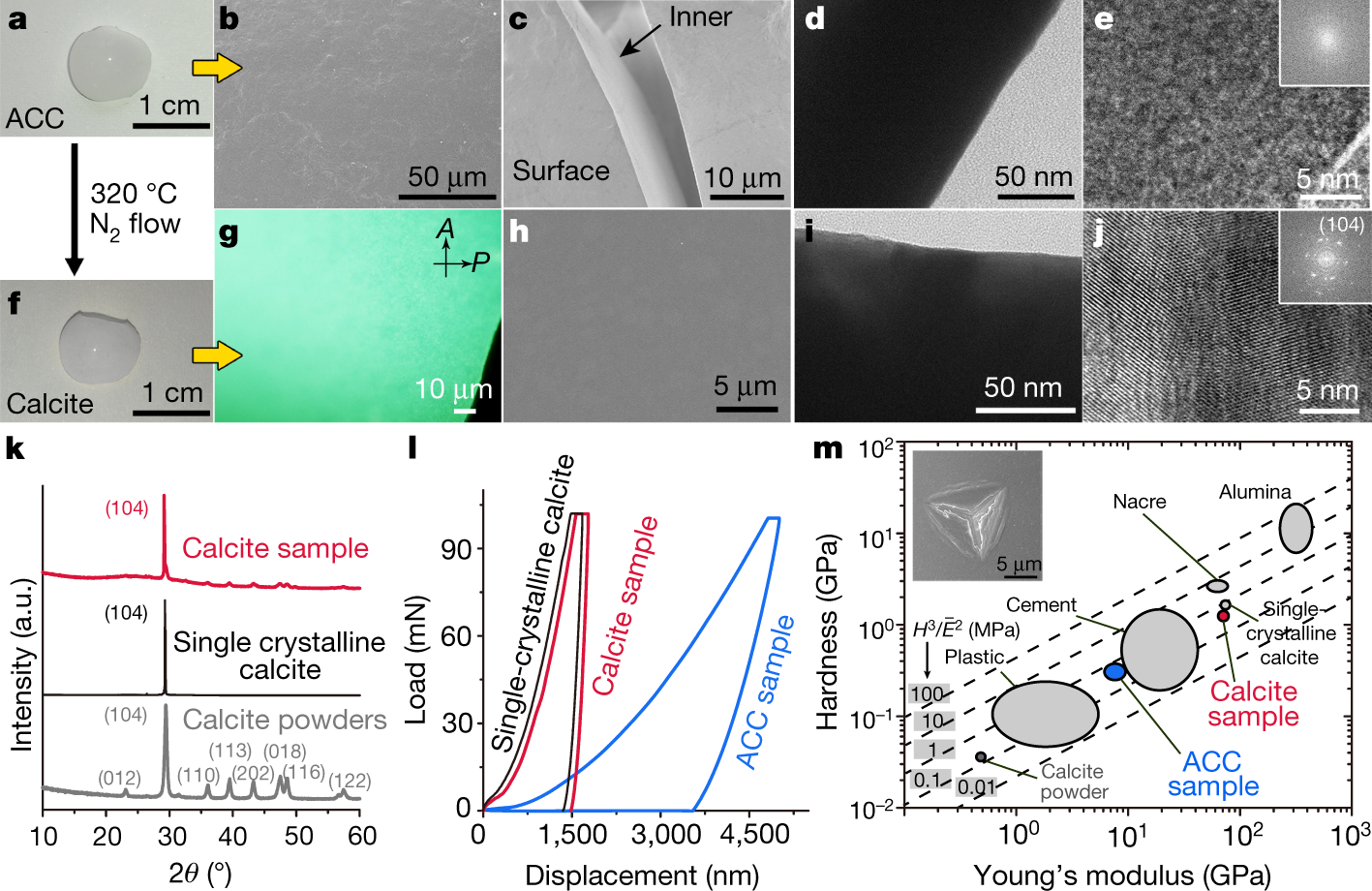
The caption:
ACC, is amorphous calcium carbonate.
Some discussion of possible applications:
A graphic on engineering utilizing this technique:
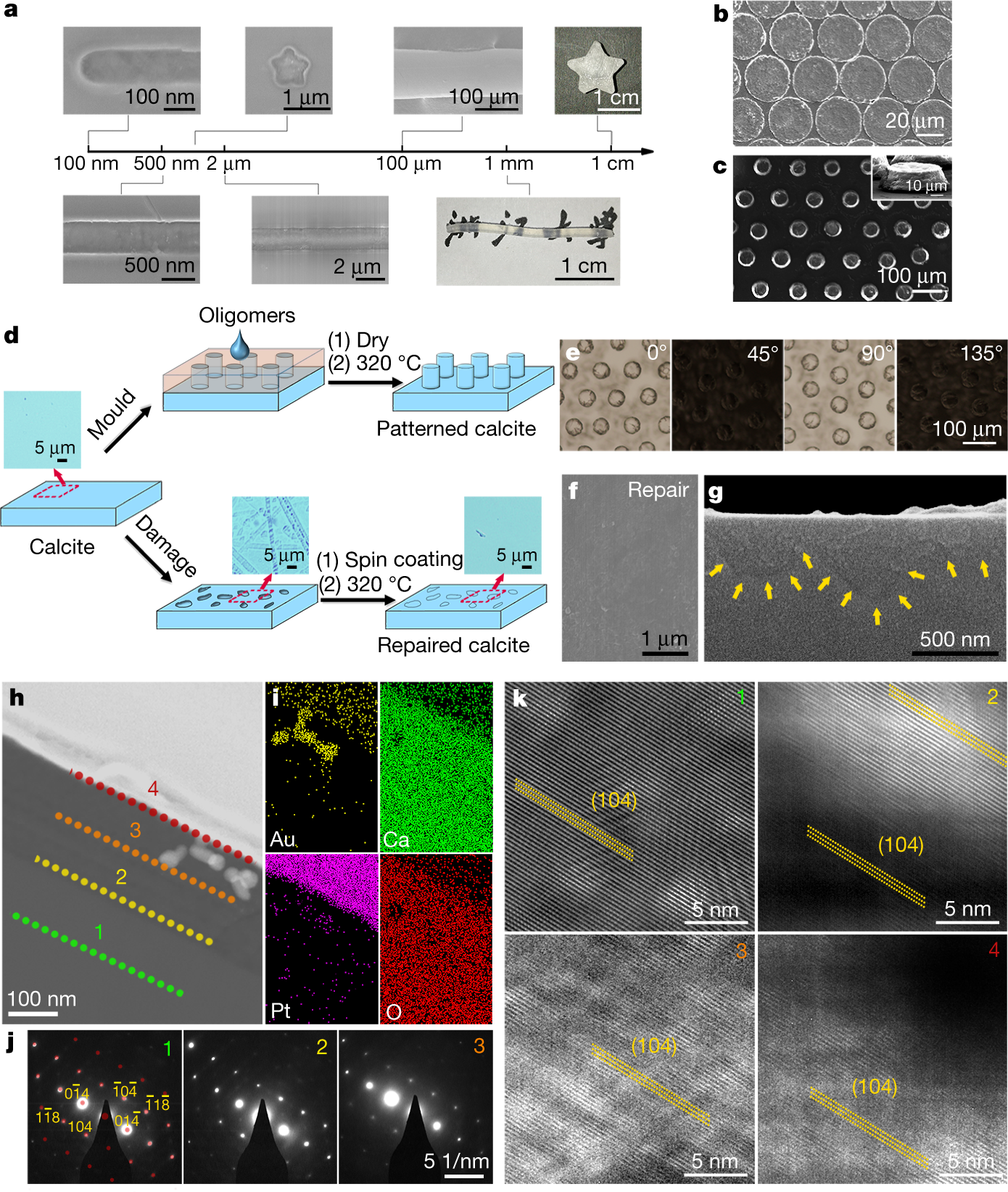
Interesting paper, I think, I thought I'd share it.
Have a great Friday.
Milk of ruminants in ceramic baby bottles from prehistoric child graves
The paper I'll discuss in this post is this one: Milk of ruminants in ceramic baby bottles from prehistoric child graves (J Dunne et al Nature 574, 246–248 (2019)).
It is, I think, a good thing that we found out that the most modern approach to infant nutrition is breast feeding. Chemical analysis of human breast milk has shown - aside from chemical toxins associated with our modern life style, for example the common presence of brominated diphenylether flame retardants in the women who recycle our electronic stuff in China - that it is unique among all animal species. One of the major differences concerns lipids; human breast milk contains for example certain polyunsaturated lipids such as EPA, eicosapentenoic acid, and the related DHA and aracadonic acid which is thought to offer certain developmental neurological advantages owing to their propensity to undergo cycloadditions to make important molecules such as prostaglandins and certain hydroxylated species. I'm not sure that I know the precise mechanisms of the neurological effects, but I do know they are real.
I used to joke with my wife that I hoped that my sons would wean before they went to college; I'm sure both of them breast fed at least past their third birthday, although they ate other foods before their first birthday. They're men now, and it's possible, I think, it all worked out for the better. They're pretty smart guys.
Anyway, it appears that the problem of feeding children nonhuman animal milk has a very long history according to this paper. From the abstract which is open sourced:
From the introduction to the paper, which is probably not open sourced:
Breastfeeding is integral to infant care in all human groups and fundamental to the mother–infant relationship4. Breast milk provides an infant with all of the macro- and micronutrients that are required to sustain growth for the first six months of life9, together with bioactive components, which protect the infant from pathogenic organisms and facilitate the development and maturation of the immune system10. The introduction of energy and nutrient-rich, easily digestible, supplementary foods in infant feeding (that is, during weaning) is unique to humans11,12. Supplementary foods are generally introduced at around six months of age, when the metabolic requirements of an infant exceed the energy yield that the mother can provide through milk, contributing to the infant diet as chewing, tasting and digestive competencies develop1,12,13.
Apparently vessels that are believed to have been ceramic "baby bottles" were placed in the graves of children buried in prehistoric Germany:
Possible infant-feeding vessels that are made from clay first appear in Neolithic Europe. One of the earliest of such finds is a Linear Pottery Culture feeding vessel from Steigra, Germany, that has been dated16 to around 5500–4800 BC. These unique vessels, which have a small spout through which liquid could be poured or suckled, come in many forms and sizes and occasionally have a zoomorphic design (Extended Data Fig. 1). They become more common in Central Europe during the late Bronze and early Iron Age4 and are found in settlements, as stray finds, and in graves (particularly those of children), which strongly suggests that they were feeding or weaning vessels for infants.
The precious nature and often small openings of these vessels makes their sampling for organic residue analysis extremely challenging. However, infant-feeding vessels that have an open, bowl form, found in graves from cemeteries of Dietfurt-Tankstelle and Dietfurt-Tennisplatz in Germany, have recently become available for chemical analysis. The graves are part of a large early Iron Age cemetery complex (dating to approximately 800–450 BC) found in the lower Altmühl valley in Bavaria, Germany, with Dietfurt-Tankstelle encompassing 99 burials in 72 graves17 and Dietfurt-Tennisplatz containing 126 burials18. Child grave 80 at Dietfurt-Tennisplatz contained an east–west-oriented inhumation of a young child (0–6 years old), who had a bronze bracelet on the left arm, and in which feeding vessel 1 (Fig. 1a) was placed at the child’s feet18.
A graphic:
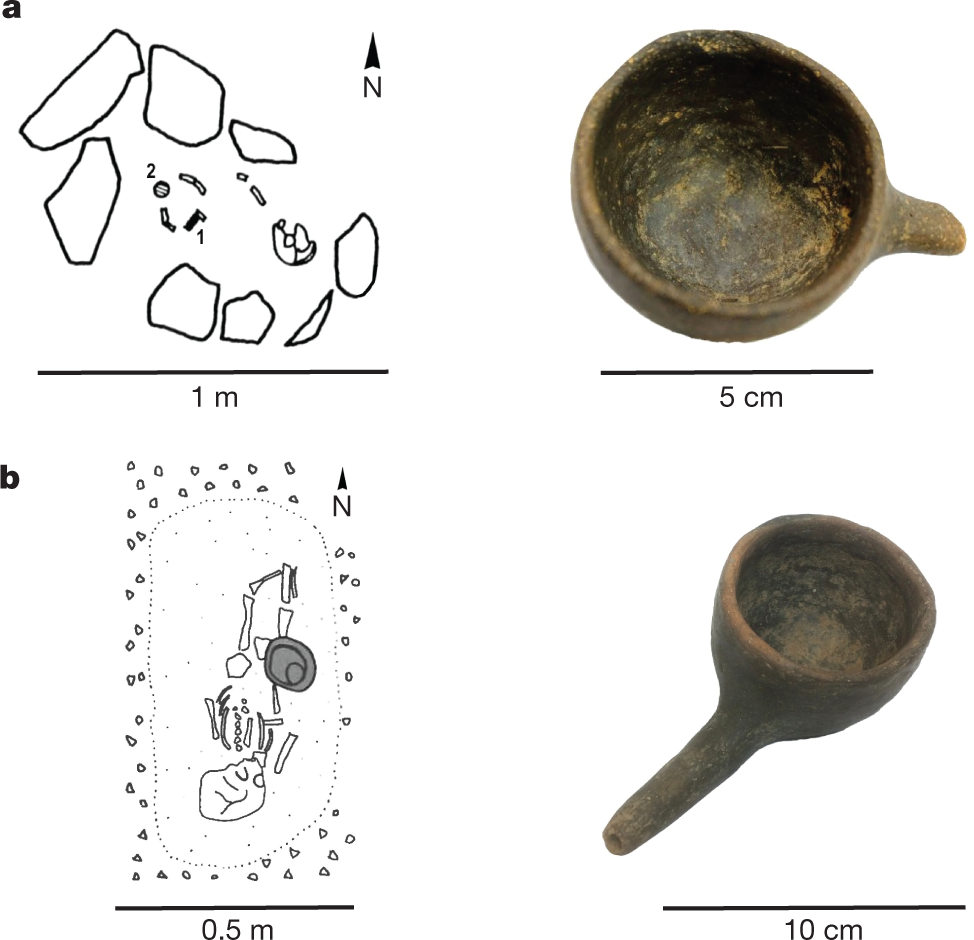
The caption:
In the labs where I work, we analyze fatty acids by LC/MS/MS which is generally more sensitive than GC-MS single quads with derivatization to FAMES (fatty acid methyl esters), but the authors here have used the latter, and apparently it got the job done, utilizing the NIST databases. (I'd personally be reluctant to do this with samples this precious, but it worked out apparently; the paper is published in Nature.)
Their results:
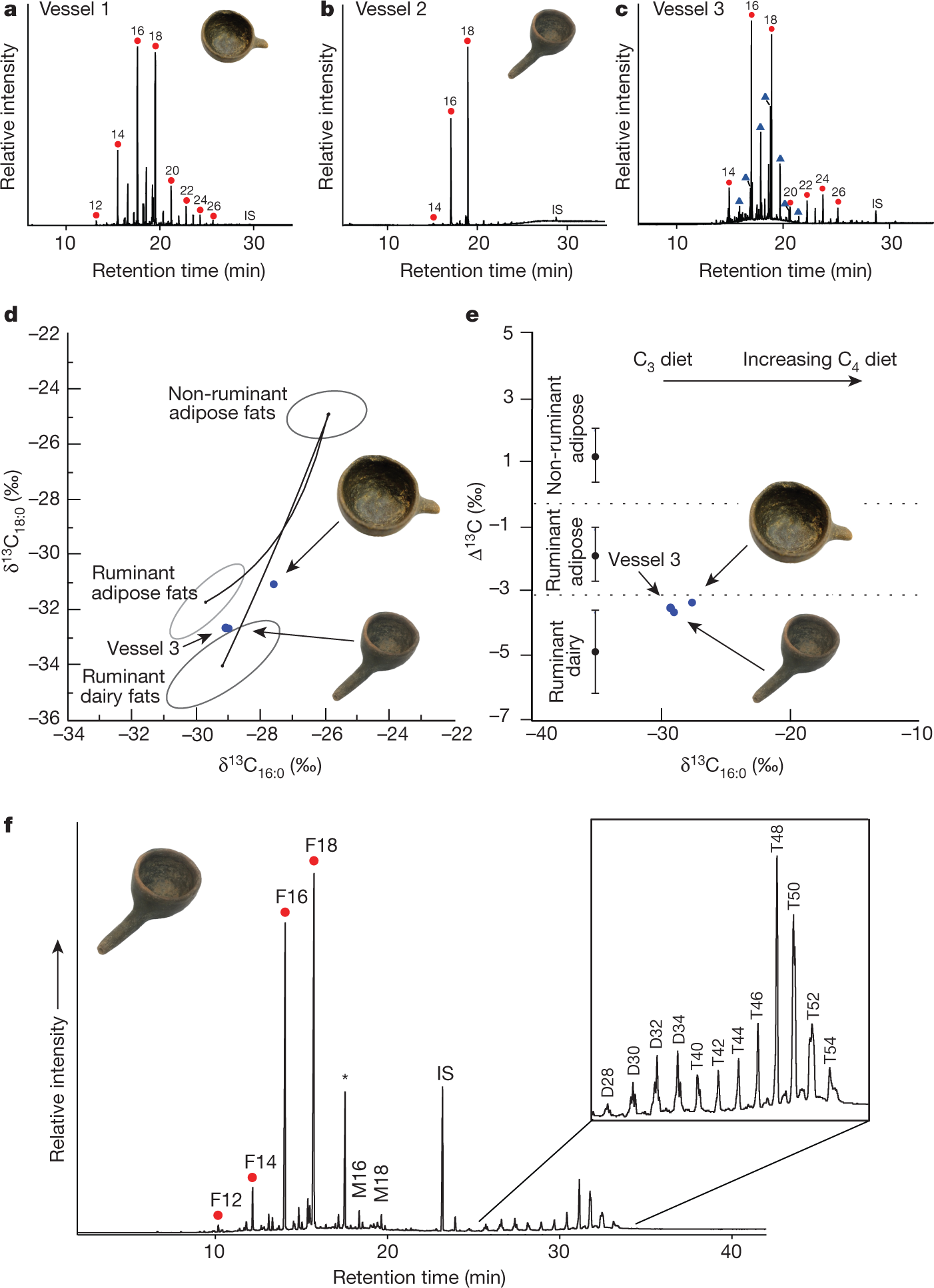
The caption:
Here's some explanation of the different lipid profiles of cow's milk and human milk:
Nutritional differences:
My sons were both with me this weekend, and we had this fascinating conversation on why European culture came to be so widely adopted across the world, which of course, is tied to imperialism, the latter being tied to a cultural propensity for violence that was not absent in other cultures, but seems to have been less intense, the Mongols, perhaps excepted. I mentioned Kurt Vonnegut's description of Europeans as "sea pirates" which other cultures refused to believe could possibly be as greedy and violent as they turned out to be. My younger son had an interesting theory related to geography to explain the warlike practices of Europeans. We were just kicking stuff around. I don't know that we were being cartoonish; I'm sure we were.
This may only be loosely tied to nutritional differences, but these nutritional differences have had an effect on culture.
It is well known for example, from genetic data, that Europeans as distinct from other ethic groups exhibit more tolerance for lactose than other ethic regions, which is why one never sees cheese sauce for example, in Chinese or Japanese food. It is also well known that, albeit, with many exceptions, that certain cancers are less prevalent in Asia, notably breast cancer.
Seemingly small things matter.
In any case, I found this interesting chemoarcheology to be very interesting, and I thought I'd share it.
Have a nice day tomorrow.
Joan Brennecke Returns in Triumph to her Alma Mater with a 2.5M Grant.
Whenever I see a paper from Joan Brennecke while growing through titles in a journal I just have to read it. (Confession: The Journal she edits, Journal of Chemical Engineering Data, is not on my regular reading list; I wish I had more time.)
Most recently I came across this paper of hers: Effect of Water on CO2 Capture by Aprotic Heterocyclic Anion (AHA) Ionic Liquids (Gabriela M. Avelar Bonilla, Oscar Morales-Collazo, and Joan F. Brennecke, ACS Sustainable Chem. Eng. 2019, 7, 16858?16869)
I usually don't notice institutions, but it caught my eye, that after a long career at Notre Dame, she is now at the University of Texas at Austin. So I googled to find out what happened and came across this article: Engineering a New Era
Joan Brennecke, the first female professor of chemical engineering at the University of Texas, has charted her own career course.
As a chemical-engineering student at the University of Texas in the 1980s, Joan Brennecke learned more than formulas and chemical processes; she learned how to stick up for herself. Some professors supported Brennecke in her engineering ambitions, helping her become more assertive and self-assured. Meanwhile, others represented the challenges she would face throughout her career in a male-dominated field.
Brennecke remembers casually chatting with a UT chemical-engineering professor about her career ambitions at a party in 1984, when he surprised her with a mocking laugh.
“A female faculty member in chemical engineering at the University of Texas? Over my dead body!” he declared.
After a $2.5 million governor’s grant returned the world-class researcher to her alma mater three decades later, Brennecke can’t help but laugh at the story’s irony.
“I try never to whine about others’ behavior,” says Brennecke, now UT’s first female full professor in chemical engineering. “I tend to ignore it and do my thing.”
Engineered to Succeed
Brennecke knew chemical engineering was her thing since early high school. She recalls hours spent in the garage with her father, a chemical engineer with a Ph.D., taking apart anything the two could find. When she was 12, they disassembled a massive mechanical calculator he brought home from his job at Alcoa.
Engineering runs in the family: Brennecke’s uncle works as a mechanical engineer, her mother is a secretary for an engineering company and three cousins ended up in engineering-related positions. Brennecke entered the family hall of fame as its first female engineer.
Today, women earn about 20 percent of all engineering degrees. When Brennecke hit high school in the mid-1970s, women earned just 3.4 percent of those degrees...
...Gabriela Avelar Bonilla, one of Brennecke’s Ph.D. students at Notre Dame, says Brennecke serves as a valuable resource for women in engineering.
“In a field that [is]usually dominated by men, it’s important to have role models that you can relate to,” Avelar Bonilla says, “[especially]someone like her because her career is very impressive and she is a good mentor.”
When paired with a female mentor, female engineering undergrads feel more confident, motivated and less anxiety, according to a study published earlier this year.
The first piece of advice Brennecke offers female engineering students is to focus on doing their best.
“There’s no substitute for competence,” she says.
“And for goodness’ sakes, don’t ever be dissuaded or even irritated by somebody’s stupid comments or what somebody does. Don’t waste your brain cells on them. Spend your brain cells on doing what you’re doing well.”
Dr. Brennecke gave a wonderful lecture at Princeton University which I attended a few years back. I have posted the video in this space here:
On the Solubility of Carbon Dioxide in Ionic Liquids.
I was, at the time, a little depressed about the remarks on SO2, but the world has gone beyond that.
In any case, it's great to see powerful women in Chemical Engineering. Dr. Brennecke is a national asset.
Diagramming making bad coffee.
I was reading a paper today, this one, Development and Optimization of Liquid Chromatography Analytical Methods by Using AQbD Principles: Overview and Recent Advances and I came across a reference to a "Ishikawa Fishbone Diagram."
We are fortunate in these times that when we don't know what something is, we can google it, and often end up at Wikipedia, which is what happened to me, where I learned how to make a diagram of how I might make bad coffee:
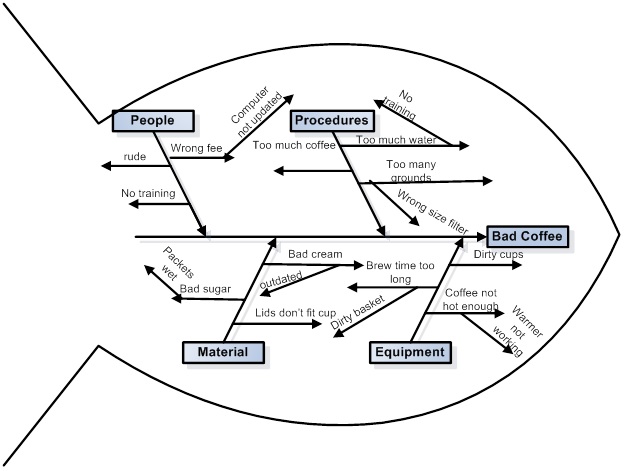
Feel free to follow these steps to making bad coffee.
It turns out that I've seen these types of diagrams before, but never knew what they were called. Now when I'm in a meeting, I can say "Ishikawa Fishbone Diagram" and sound like I know something, even if I don't know shit from shinola.
I was mistaken about the timing and magnitude of the 2019 Mauna Loa CO2 minimum.
On September 22 I wrote the following in a post in this section:
This year, in 2019, as is pretty much the case for the entire 21st century, these minima are uniformly higher than the carbon dioxide minima going back to 1958, when the Mauna Loa carbon dioxide observatory first went into operation. Weekly data is available on line, however, only going back to the week of May 25, 1975, when the reading was 332.98 ppm.
For many years now, I have kept spreadsheets of the data for annual, monthly, and weekly Mauna Loa observatory data with which I can do calculations.
In the weekly case, the week ending May 12, 2019 set the all time record for such readings: 415.39 ppm.
These readings, as I often remark vary in a sinusoidal fashion, where the sine wave is imposed on a monotonically increasing more or less linear axis, not exactly linear in the sense that the slope of the line is actually rising slowly while we all wait with unwarranted patience for the bourgeois wind/solar/electric car nirvana that has not come, is not here and will not come.
This graphic from the Mauna Loa website shows this behavior:

Here is the data for the week beginning on September 15, 2019
Up-to-date weekly average CO2 at Mauna Loa
Week beginning on September 15, 2019: 408.50 ppm
Weekly value from 1 year ago: 405.67 ppm
Weekly value from 10 years ago: 384.59 ppm...
...The operative point is that this reading is only 0.09 ppm lower than last week's reading, which was, 408.59 ppm. This suggests, if one is experienced with working with such data, that this is most likely the annual September minimum reading. For the rest of this year, and through May of 2020 the readings will be rising. We will surely see next May readings around 418 ppm, if not higher.
However, I was wrong, because the next two weeks at Mauna Loa showed values lower than 408.50 ppm. It actually took place this year during the week ending 09/29/19, when the reading was: 407.97
The most recent data point is the week ending October 6, 2019 is a follows:
Up-to-date weekly average CO2 at Mauna Loa
Week beginning on October 6, 2019: 408.39 ppm
Weekly value from 1 year ago: 405.50 ppm
Weekly value from 10 years ago: 384.06 ppm
Last updated: October 13, 2019
From here on out, until May, 2020, the values for each week will exceed the number reported on September 29 of this year.
Previous weekly data annual lows took place as follows over the last 5 years:
9/9/18: 405.39 ppm
9/24/17: 402.77ppm
9/25/16: 400.72ppm
9/27/2015: 397.2 ppm
9/14/2014: 394.79 ppm
No one alive today will ever see a measurement at Mauna Loa lower than 400 ppm again.
In 2000, the weekly data annual low took place on September 10, 2000: 367.08 ppm.
In 1980, the weekly data annual low took place on September 4, 1980, 339.87 ppm.
In 1975, the first year the weekly data was reported, the weekly data annual low took place on August 31, 1975 when it was 329.24 ppm.
The movement to late September is most probably a function of a warmer and longer summer in the Northern Hemisphere, during which the annual minimums take place.
The annual maxima show up in early May. We may expect that the 2020 maximum should approach or exceed 418 ppm.
I apologize for jumping the gun. It's possible that next year we'll see, for the first time ever, the minimum appearing in October.
Have a nice afternoon.
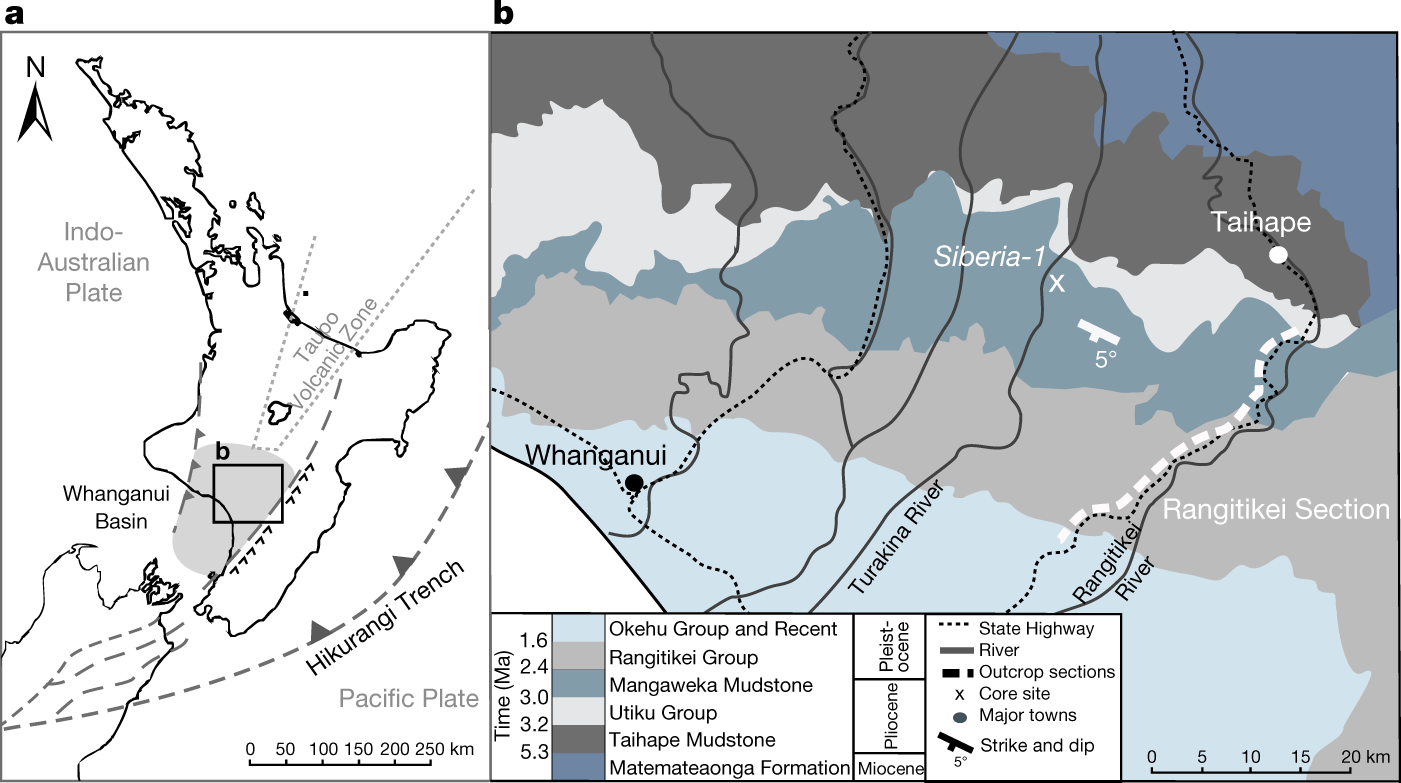
The amplitude and origin of sea-level variability during the Pliocene epoch
The paper I'll discuss in this post is this one: The amplitude and origin of sea-level variability during the Pliocene epoch (Grant et al, Nature volume 574, page s237–241 (2019).
This past Thursday I posted a similar paper about this epoch, which was also published in Nature, in the same issue, just above this one.
During the Pliocene Epoch, which was from 3 to 5 million years ago, the concentration of carbon dioxide in the atmosphere apparently surged (for a few hundred thousand years) to around 450 ppm, which, since we are doing nothing meaningful about climate change, we will hit in about 15 to 20 years.
The authors here use a different approach than the approach I discussed on Thursday.
From the abstract:
The authors review, as the authors described in my previous post, the techniques for evaluating the sea level in the geological past:
...and describe some significant limitations, for example with the ?18O method...
They then describe their approach:
Reference 6 is this one, from the same group:
Mid- to late Pliocene (3.3–2.6 Ma) global sea-level fluctuations recorded on a continental shelf transect, Whanganui Basin, New Zealand (Grant et al Quaternary Science Reviews Volume 201, 1 December 2018, Pages 241-260) I have not personally accessed this paper.
A few more details on their approach:
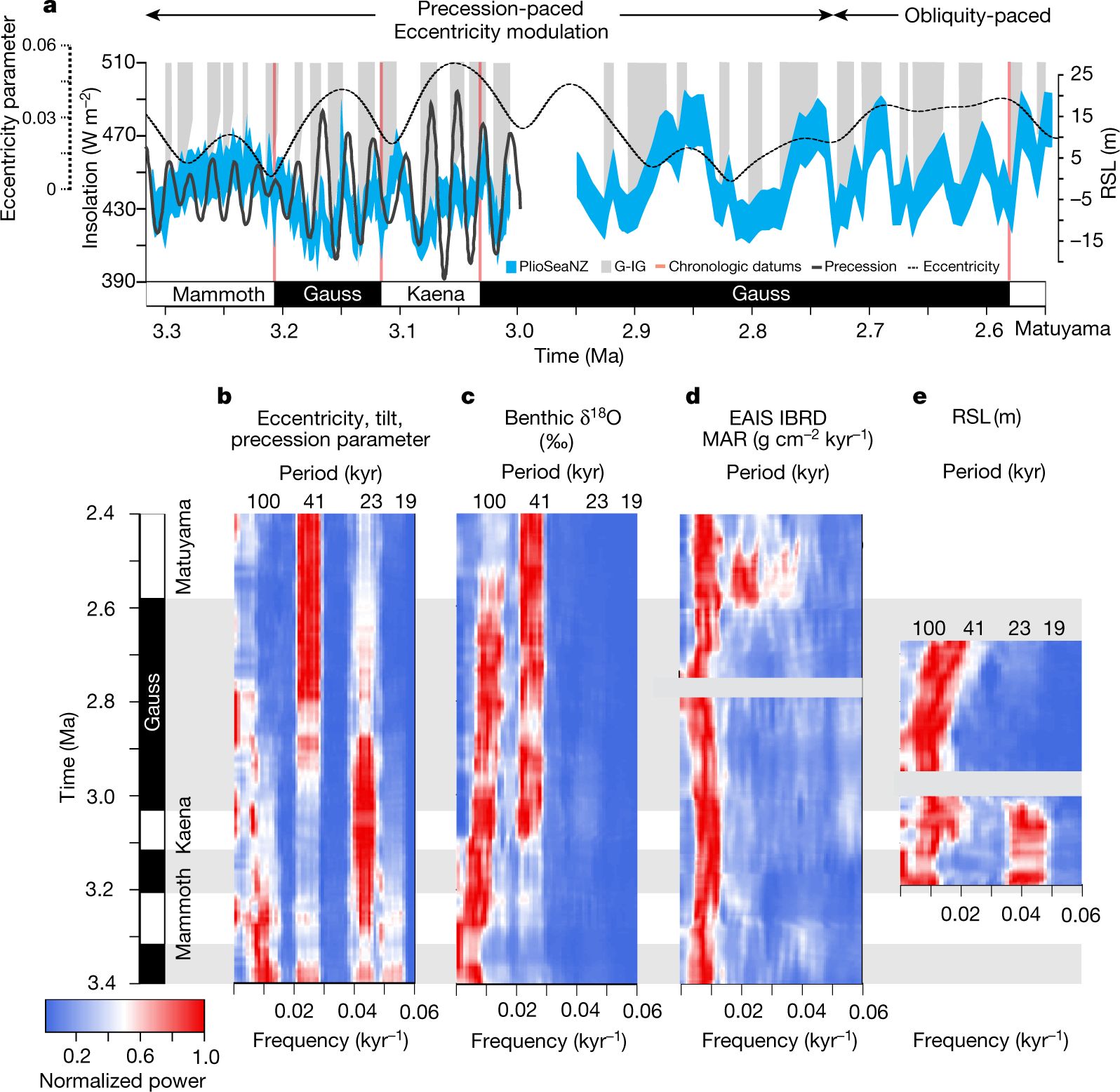
Some pictures from the text:
The caption:
 ?as=webp
?as=webp
The caption:
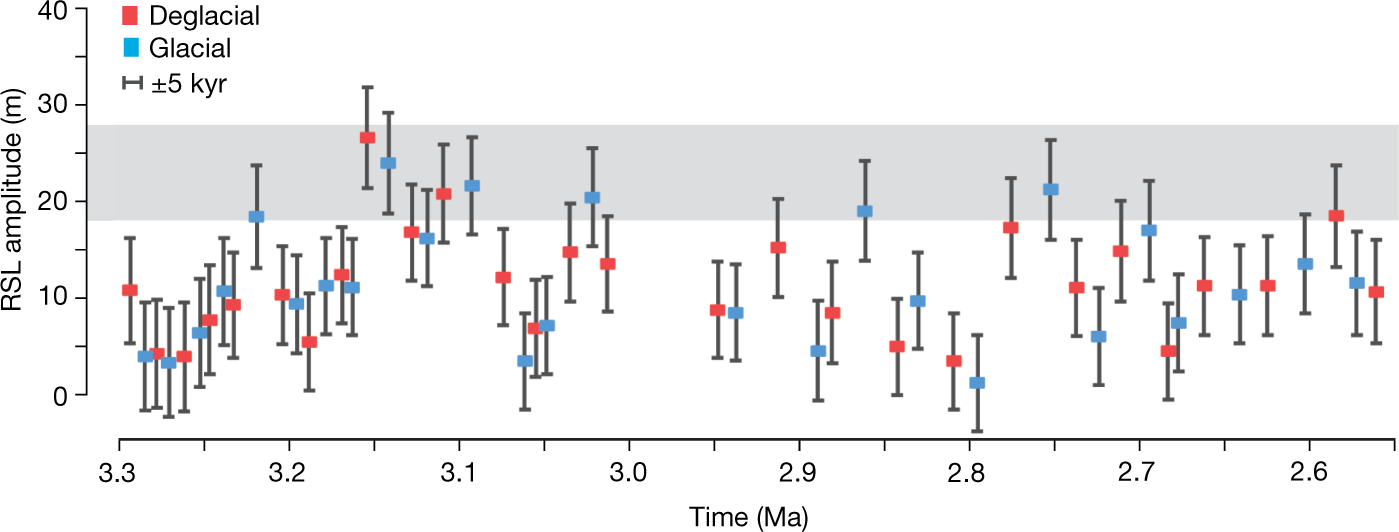
The caption:
https://www.nature.com/articles/s41586-019-1619-z/figures/4
The caption:
AIS is the antarctic ice sheet, GIS, greenland ice sheet.
They speak on the effect of rotational precession changes the insolation patterns drive this historical warming, and that this in turn, they argue, means that the Antarctic Ice Sheet is more prominent in driving sea level rises.
This does not mean that they exclude carbon dioxide, far from it.
From their conclusion:
Have a pleasant Sunday afternoon.
A Tale of 2 Radioactive Contamination Issues: the San Joaquin Oil Fields & Fukushima Seaweed & Tuna.
The two papers I will discuss in this post are from a recent issue, as of this writing, of one of my favorite scientific journals, Environmental Science and Technology.
They are:
Occurrence and Sources of Radium in Groundwater Associated with Oil Fields in the Southern San Joaquin Valley, California (McMahon et al, Environ. Sci. Technol. 2019, 53, 9398?9406.
...and...
Temporal Variation of Radionuclide Contamination of Marine Plants on the Fukushima Coast after the East Japan Nuclear Disaster (Arakawa et al Environ. Sci. Technol. 2019, 53, 9370?9377)
I will also briefly discuss, in papers linked below, the famous Fukashima Tuna Fish.
The second paper is behind a firewall but may be accessed in an academic library; the first is open sourced and anyone can read it.
For convenience I will treat them both as if they were behind a firewall, and excerpt portions and graphics of both.
Before launching into a discussion of the papers, it is worthwhile to discuss the nuclear properties of all the species discussed in the paper. To do this, I will provide references to other papers as well as links to those that have them. These papers have largely been downloaded or scanned into my personal files but many, if not all, may be accessed in a good academic library.
Almost all of the data in the discussion of the nuclear properties here were sourced from the website of the National Nuclear Data Center by entering the symbol for the nuclides in question in the top box and clicking the "decay data" in the right hand box. This will lead to other links, where one can use the "human readable" form to get the information on half-lives and decay energies. This data can also be accessed using the "periodic table browse" tab.
The radioactive nuclides discussed in the first paper are all anthropogenic nuclei created in the operation of nuclear reactors, two isotopes of cesium-134 (Cs-134), and cesium-137(Cs-137). The latter is a fission product. The former is formed by neutron capture in non-radioactive cesium-133, also a fission product. (The direct formation of cesium-134 does not take place since Xenon-134, a fission product preceding it is observationally stable and thus not subject to measurable radioactive decay.) Also discussed is an isotope of silver, silver-110m (Ag-110m), a nuclear isomer of radioactive silver-110 (Ag-110). Cs-134 has a half-life of 2.06 years. Cs-137 has a half-life of 30.08 years. Ag-110m has a half-life of 249 days, roughly.
It has been roughly 3100 days since the last reactor at Fukushima suffered a hydrogen explosion. The fraction of Ag-110m that remains is about 0.000178 of that which was present when the explosions took place, and, as silver chloride is insoluble, I will not discuss this isotope at any length in this post even though it mentioned in the paper on Fukushima to be discussed. It is however discussed in the paper as a kind of marker, despite it's extremely low concentrations.
The decay energy of Cs-137 is nominally 594 keV, however most of this energy is carried off as a neutrinos with a mean energy of 370 keV. As neutrinos interact only weakly with matter there is little effect on matter, living or dead. Most of the energy that does interact with matter is in the form of a ?- particle, with a mean energy of 179 keV. Beta particles are not particularly penetrating, but can damage local matter when it decays, including living tissue if it is in contact with it or internalized in it.
Cesium-137 is, however, responsible for two nuclear decays, that of itself, and that of its daughter, Ba-137m, the nuclear isomer of stable Ba-137. Since the half-life of Ba-137m is 2.55 minutes, Cs-137 is in secular equilibrium with Ba-137m, which emits a 594 keV gamma ray in its isomeric transition, under most conditions. Since the decays are quite nearly simultaneous on an effective transport time scale, this gamma ray is sometimes reported as the decay energy, which is 661 keV (0.661 MeV) of cesium although it is actually a decay in barium. The concentration of Cs-137 is thus often determined by the secular equilibrium ratio by detection of this Ba-137m gamma ray. Gamma rays can interact strongly with matter by breaking chemical bonds, even strong bonds, for example carbon-fluorine bonds. They also can easily break bonds in living tissue, which is why they can kill cells, and in high enough doses, whole organisms including human beings.
Barium-137m can be placed in disequilibrium by exploiting the insolubility of barium carbonate or sulfate. Cesium sulfate and/or carbonate are completely soluble. If disequilibrium occurs, and the mixture is not subject to additional active separation, equilibrium represented by the maximal accumulation of Ba-137m is reestablished in about 57 minutes.
(I often reflect on this disequilibrium when considering certain kinds of radiocesium hyroxide devices for storing (and/or converting) decay energy for work utilizing compressed air, since a side product of the scheme would be air capture of carbon dioxide and the destruction of certain kinds of problematic long lived greenhouse gases that have been released into the atmosphere by the refrigeration industry.)
Cs-134, the other radioactive cesium isotope released in significant quantities at Fukushima, decays directly to stable Ba-134. It has a decay energy of 2,058 keV (2.058 MeV), much of it released in the form of an gamma rays with an average energy of 1558 keV (1.558 MeV) It decays almost exlusively by ?- emission; the average energy of the ?- particle is 157 keV.
Ag-110m largely decays directly to cadmium-110. It has a decay energy of 2,968 keV (2.968 MeV). Most of it decays by ?- emission, with an average(? emitter, HL seconds, decay energy, keV ( MeV)) particle energy of 67 k and a gamma ray with an average energy of 2,760 keV, (2.76 MeV). A small amount (1.36%) decays to Ag-110, which has a half-life of around 24 seconds and a similar energy to the parent 110m nuclear isomer.
All three of these isotopes, Cs-137, Cs-134 and Ag-110m were released by the meltdown of the Fukushima reactors.
The paper discusses the fate of these isotopes in seawater and in seaweed in the general Fukushima area, as well as their relative concentrations.
The radioactive species discussed in the second paper are all called Naturally Occurring Radioactive Materials (NORM). It is chiefly concerned with two of these, radium-226, a part of the uranium-238 decay series, and Radium-228, a part of the thorium decay series. Both are highly energetic alpha emitters. Ra-226 is the parent isotope of 9 (or more, depending on branch ratios) additional nuclear decays. Ra-228 is the parent leading to 10 additional nuclear decays.
The half-life of Ra-226 is 1600 years. The half-life of Ra-228 is 5.75 years.
The decay energy of Ra-226 (? emitter, HL 1600 years, decay energy, 4,871 keV (4.871 MeV)) is almost an order of magnitude higher than the decay energy of Cs-137, the longest lived, and in many ways the most problematic of all fission products. The daughter nuclides of the decay of Ra-226 are these, with "Half-life" abbreviated "HL:"
Rn-222 (? emitter, HL 3.8235 days, decay energy, 5,590 keV (5.590 MeV)),
Po-218(? emitter, HL 3.098 minutes, decay energy, 6,114 keV (6.114 MeV)) (minor 0.02% ?- ),
Pb-214 (?- emitter, HL 26.8 minutes decay energy, 1,019 keV (1.019 MeV)),
Bi-214 (?- emitter, HL 19.9 minutes decay energy, 3,270 keV (3.270 MeV)) ,
Po-214(? emitter, HL 164.3 ?seconds, decay energy, 7, 833 keV (7.833 MeV)) ,
Pb-210 (?- emitter, HL 22.2 years decay energy, 63.5 keV (0.0635 MeV)) ,
Po-210 (? emitter, HL 138.376 days, decay energy, 5,407 keV (5.407 MeV)),
Pb-206 (stable)
The reason for producing these details is to show that the energy associated with the decay of radium is much higher than the energy associated with the decay of the radioactive cesium isotopes. If this energy is deposited in living tissue the matter is more serious.
The effects of radiation on tissue is the subject of efforts to systematize it as so as to make reasonable assessments of risk; a considerable effort has been made which is probably very good to a first approximation to produce something called “quality factors” which is a function of the type of radiation, in most cases here alpha radiation, which is not very penetrating and thus deposits most of its energy locally, the type of tissue and the density of the tissue in which it traveling. This is the type of thinking that goes into the unit “Sievert,” abbreviated “Sv” which is frequently a unit mentioned with respect to the health risks of radiation. It has replaced the “Rem” in common radiation health, and accounts for the type of radiation.
A related unit is the “Gray” which is a measure of the amount of energy deposited in a material, which is also a function of the nature of the material, extending beyond tissue. The Gray involves less subjectivity than the Sievert.
The total amount of energy generated by the decay of radium and it’s daughters, in their decay is a whopping 34,186 keV. Since much of this energy is in the form of low penetrating ?- and ? particles, it follows that much of it is in fact deposited in tissues if the nuclide is internalized in the tissue.
The unit keV is an atomic unit and may not mean much to the average citizen not accustomed to dealing with such units. The conversion factor between this unit and the more familiar energy unit the Joule is simply the charge on an electron, 1.609 X 10^-19 Coulombs times 1000 to account for the “k” from kilo-. It is useful to consider how much energy a gram of “radium daughters.”
Of the above nuclei listed in the radium decay series, the one with the shortest half-life that is actually long lived enough to have actually been isolated is Po-210. This is the nuclide that Vladmir Putin’s agents utilized to kill the renegade Russian Security Agency Defector Alexander Litvinenko. (All of the world’s commercial Po-210 is manufactured in Russia and is exported to countries around the world to control static in the manufacture of very sensitive microcircuits for example.) Therefore to consider how much energy a “MeV” is, let’s take a look at the macroscopic energy output of a gram of this element’s isotope puts out. Using the conversion factor from the previous paragraph, we see that the decay of a single atom of Po-210 puts out .870 nanoJoules of energy, or 870 picoJoules.
The number of atoms decaying in a second is given by the specific activity of Po-210, which is in turn, determined by its decay constant, ?, which is defined by dividing the natural logarithm of 2 by the half-life in seconds, which in the case of Po-210, works out to 5.80 X 10^(-8) inverse seconds. Multiplying this number by Avogadro’s number gives the number of decays in a mole of Polonium-210, and dividing it by the approximate atomic weight of Po-210, 210, gives the number of decays per second. This number is 2.5 trillion decays per second per gram. Multiplying this by the decay energy, we see that a gram of Po-210 puts out about 2,170 watts of power.
The density of the beta phase of polonium, which is the only phase that can be reliably measured given the heat output of the metal, is about 9.4 g/ml, meaning that the volume of a gram is simply the inverse of this number or about 0.11 ml. A teaspoon is said to be 9.2 ml, and it follows that a gram of polonium-210 putting out 2,170 watts is about 0.02 teaspoons.
It is the high energy to mass ratio that makes nuclear energy environmentally superior to all other forms of energy.
However, as is clear from the Litvinenko case, ingesting or being injected, even in amounts much less than a gram, of Po-210 will kill a person.
Much of what I have written above is misleading, in the sense that it implies that if one were to eat radium, or to drink it in well water contaminated with it – the possibility of which is discussed in one of the papers, to be discussed herein – one would be subjected to all of the decays during one’s lifetime, which is decidedly not true. A “bottleneck” in the decay series is the relatively long half-life of lead-210. The radioactive elements in a decay series are subject to various kind of equilibria, and the equations of radioactive decay, derived from the Bateman Equation.
From the use of these equations, it can be shown that the attainment of transient equilibrium between Ra-226 and Pb-210 would take about 139 years to achieve, at which time the ratio between the parent nuclide (Ra-226) and the daughter (Pb-210) would be 0.0139. Moreover the equilibrium ratio of Pb-210's daughter, Po-210, to Pb-210 would be 0.017, meaning that the ratio of ratio of Po-210 to Ra-226 would be 0.00027 or 0.027%. Only 94.2% of the radium originally present would remain at this point.
This is why Marie Curie was not immediately killed by isolating radium; it took a long time to reach Po-210 equilibrium. In fact, it took 36 years for her work with radium (and other radioactive elements) to kill her, a period of remarkable scientific achievement during which she was awarded two Nobel Prizes. (She and her husband had previously isolated tiny amounts of Polonium, before their discovery of radium, from uranium ores, but not enough to kill them; the amounts were invisible, and in fact, still shielded by significant quantities of uranium.
But let’s be clear, her work with radioactivity did kill her.
Nevertheless, people do regularly consume polonium, at least to the extent they eat seafood.
The ocean contains about 4.5 billion tons of uranium, albeit in low concentrations, a generally accepted average level being around 3.3 ?g per cubic meter, although this figure can vary from place to place. In the Mediterranean Sea, for example, measurements of concentrations of uranium in seawater ranged from between 3.2 to 3.7 ppb, (XAS and TRLIF spectroscopy of uranium and neptunium in seawater Melody Maloubier, et al Dalton Trans., 2015,44, 5417-5427), between 3.01 and 3.15 ppb in the anoxic Saanich Inlet, a fjord on Vancouver Island in Canada (Uranium isotope fractionation in Saanich Inlet: A modern analog study of a paleoredox tracer (C. Holmden et al. Geochimica et Cosmochimica Acta 153 (2015) 202–215) and fairly precise measurements in the seas around Tiawan gave 3.116 ppb with a relative error of around 1.6%. Measurements of natural uranium concentration and isotopic composition with permil‐level precision by inductively coupled plasma–quadrupole mass spectrometry (Shen et alGeochem. Geophys. Geosyst., 7, Q09005, )
Uranium in seawater has been there ever since the Earth's atmosphere began to feature significant concentrations of oxygen, for billions of years. It has thus had plenty of time to come into transient equilibrium with its daughter radium. It takes "only" 34,282 years for this equilibrium to be established. Although radium carbonate and radium sulfate have low solubility products, they are high enough that disequilibrium is not obtained commonly in seawater; it is estimated that the scavenger lifetime of radium in seawater is six times longer than its radioactive half-life. (cf Teh-Lung Ku and Shangde Luo, U-Th Series Nuclides in Aquatic Systems, Cochran and Krishnaswami, ed. Ch 9 pg 313).
It follows that seawater naturally contains Po-210, and, it turns out, organisms concentrate this polonium.
A rather famous paper in the scientific literature, famous mostly for the idiocy which the international media interpreted it, concerns the "Fukushima Tuna Fish." The paper is here: Pacific bluefin tuna transport Fukushima-derived radionuclides from Japan to California (Madigan et al PNAS 109 24 9483–9486 (2012)). The purpose of the paper was to utilize a particular marker, the relatively short-lived nuclide Cs-134, that was injected into the sea by the destruction of the Fukushima nuclear reactors by a natural catastrophe to trace the migration of tuna and not to publicize a huge health risk.
However, because of the publicity the paper generated, the embarrassed authors felt to publish a follow-up paper, Evaluation of radiation doses and associated risk from the Fukushima nuclear accident to marine biota and human consumers of seafood (Madigan et al PNAS 110 26 10670–10675 2013).
They wrote:
More than 1100 newspapers...
The journalists engaged in this reporting remind me of a really, really stupid guy who made it to my ignore list here who once represented that the collapse of a tunnel at the Hanford Reservation, which turned out to contain some slightly radioactive discarded chemical reactors, was somehow more important than the complete destruction of the planetary atmosphere by dangerous fossil fuel waste.
No wonder ignorance is accelerating climate change. There are actually people who think we cannot use the nuclear tool – the only tool which has worked on a significant scale – to address, slow, and perhaps even halt, the ongoing destruction of the entire planet’s atmosphere because some fool on a Trumpian intellectual level heard about a small tunnel with some rail cars in it with old chemical reactors contaminated with a small amount of residual plutonium. One hears these kinds of things, but one really doesn’t want to believe it.
As for the tuna fish, the weighted absorbed dose from the radiation from natural Polonium-210 in the "Fukushima Tuna Fish" was found to be 558 ?Sv from Polonium-210, 12.7 ?Sv from the natural radioactivity associated with potassium (K-40) - both tuna fish and humans would die without mildly radioactive potassium - 0.5 ?Sv from Cs-137, most of which resulted from nuclear weapons testing in the 20th century, and 0.4 ?Sv from the Fukushima Cs-134 that was being utilized to track the tuna fish migration patterns.
What is interesting in terms of actual radioactive decays, the number of decays attributed to Cs-134 in a kg of dry "Fukushima Tuna" is 4 Beq, and Po-210 only about 20 times larger, 79 Beq, as compared to the factor of risk as measured in ?Sv, which is almost 1400 times higher for Po-210 with respect to Cs-134, although these risks are almost vanishingly small in any case. Very few people get radiogenic cancer from eating the polonium in a kg of tuna fish. The radiation associated with a tuna fish is a fraction of normal background radiation and, in fact, if the tuna has spent a significant time in a can, most of the polonium will have decayed away to lead.
The point here, of all the diversion, is simply – an issue addressed by the unit “Sievert” – is to reify the fact that in terms of health, the issue is not how many decays a radioactive substance undergoes and also where it is located. A committed dose represents an ingested radionuclide, Polonium from a tuna fish for example.
The biochemistry of the radioactive elements is also very important in determining risk. The measured concentration of natural polonium in seawater is rather low, a Malaysian paper on the subject describing what the authors regard as high concentrations of the element reports concentrations of Polonium in seawater that is orders of magnitude lower than what the exasperated authors of the "Fukushima Tuna Fish" paper reported in the fish. (High 210Po Activity Concentration in the Surface Water of Malaysian Seas Driven by the Dry Season of the Southwest Monsoon (June–August 2009) (Sabuti, A.A. & Mohamed, C.A.R. Estuaries and Coasts (2015) 38: 482). This is because polonium - although a metal or semi-metal - is technically chemically a cogener of oxygen and sulfur and thus can behave like these elements. Squid also concentrate polonium, and the authors of a paper discussing the radiological effects of squid consumption in Korean and Portuguese diets. (Distribution patterns of chalcogens (S, Se, Te, and 210Po) in various tissues of a squid, Todarodes pacificus (Kim et al Science of The Total Environment 392, 2–3, 25 2008, 218-224.
The behavior of polonium as a chalcogen suggests its utility in medicine. Radiation kills cells and cells that we would like to kill are cancer cells. A particular type of antineoplastic drug that is becoming increasingly subject to research is an "ADC" - an antibody drug conjugate - in which an antibody with an affinity for cancer cells is linked to a "payload," a toxic antineoplastic drug, for example pacitaxel or vincristine. Radioactive substances like technetium can also be utilized in this way, but generally the linker is not part of the antibody structure, but is rather an introduced complexation agent, introducing issues with stability and selectivity for cancer cells. (The idea behind most medicinal chemistry approaches to cancer is to use a cell cancer killing agent which will kill more cancerous cells than healthy cells, but healthy cells are invariably killed.
When I was a kid, for a while I was working on the synthesis of selenols and tellurinols of aromatic species to model certain biological enzyme behavior, and my adviser joked that perhaps we should extend our efforts to include "poloninols." It was a joke, but actually one can imagine, should this be synthetically feasible, incorporating polonium into tyrosine, cysteine or methionine residues in proteins to make a cancer cell targeting antibody that carries polonium directly into a tumor and is filtered from the blood stream by the tumor, killing it. Whether someone has explored this option, I do not know. It would be interesting to know the chemical form of polonium in tuna fish, for example if it exists in the form of polonocysteine, for example, and perhaps this has been investigated, I don’t know.
A last diversion before turning to the papers I promised to discuss at the outset:
At the outset of this post, I disingenuously suggested that the decay energy of the entire radium series would be 34,168 keV (34.1MeV). I then went on to explain that in fact, there are decay “bottlenecks” which prevent all of this energy from being deposited in entirely in a person experiencing a radium decay in their flesh, most notably the existence of Pb-210 in equilibrium with its daughter Po-210. Nevertheless, in the case where a parent nuclide decays with a much longer half-life than it’s daughter(s), it is common practice to consider the case, to a first approximation, as if the decays are effectively simultaneous, as one does in determining the presence of Cs-137 from the Ba-137m gamma ray. The half-life of radium is sufficiently longer than all of its decay daughters up to Pb-210 so that it is pretty much possible to do this, but if one lives for decades after ingesting the radium – and people do this, as evidenced by the life of Marie Curie – only a portion of the energy from the decay of Pb-210 will be available to damage living cells. The total decay energy of radium and all of its daughters up to and including Po-214 is 28,697 keV (28.6 MeV), an enormous amount of energy on a molecular scale.
Here too, however, there is a caveat. The decay series of Ra-226 through Po-214 contains, the longest lived nuclide among them, gaseous radon-222. When deciding whether all of this energy in this decay series is deposited in flesh when an atom of radium decays, one must account for this fact, the chemical nature of radon, which is an inert noble gas.
It is well known that radon gas is associated with lung cancer. Radon gas is present wherever there is a uranium formation is present; for example, I live over the Reading Prong uranium formation and I have measured radon in by basement, where, in fact, I am writing this post. Happily for me, the concentration of this radon is below what is considered an action level.
Many mass market/popular books have been written on the subject of lung cancer among Native American uranium miners who worked in the mid-20th century and their lung cancer rates, which are significantly higher than among control populations. With appeal to some published scientific literature on this case – there is a great deal of that too, besides the popular books – as well as the popular books, I have written on this subject myself elsewhere on the internet. An excerpt:
Sustaining the Wind Part 3 – Is Uranium Exhaustible?.
There won’t be a lot of books written about the 19,000 people who will die today from the dangerous fossil fuel and biomass combustion waste called generically “air pollution,” nor many shut by the health risks of the Native American coal miners in the same general region who lost their jobs this year, but that to which we pay attention says a great deal about who we are.
It is generally understood that the risk of breathing radon gas are associated with the decay daughter discussed above, including, but not limited to polonium-210 and its parent, lead-210. It is the case, however, that the physiology of radon is not quite as simple as being inhaled and exhaled in a passive manner so that the risk is associated with a series of cases where radon atoms decay while in the lungs, depositing radioactive polonium, bismuth or lead.
For one thing, radium, radon’s parent, is, like the fission product strontium, deposited in bones, as it is a cogener of calcium and, like strontium and barium, behaves very similarly to it. Thus it is relatively easy for radon gas to be trapped in this dense matrix, much as radon gas, and for that matter helium gas – all of the world’s helium is derived from atoms once trapped in rock – to be trapped in bone. Much of the energy of alpha decays appears initially as high energy recoils. An alpha particle emitted at a few MeV is traveling at relativistic speeds, that is, a significant fraction of the speed of light, and it follows that the conservation of momentum will result in the daughter nuclei formed in the decay also traveling significant distances in the material it damages. This is well known in rock. If the rock is ground into fine particles, as in a fracking operation, these particles may be propelled to the surface of the particle or beyond it. Most oil and gas (and for that matter coal) deposits were formed by the reformation of biomass over hundreds of millions of years and deposited in sedimentary rocks (or possibly metamorphic) rocks. If the formations formed from oceanic deposits, they will contain oceanic uranium, in some cases concentrated by organisms – coral, for one example, greatly concentrates uranium from seawater – or, if formed terrestrially in lakes or in forests, will form from the uranium ores weathered. Thus these formations typically have uranium impurities in them, and thus uranium daughters.
It is true that radon can travel via recoils several millimeters in bone, and thus radium deposited near the surface of the bone can propel radon atoms out of bony tissue into other tissues. Theoretically, one would think, this would make the radium be solvated in the blood stream and possibly be transferred into the air via exhalation through the lungs.
It is not, however, quite that simple.
Naively, assuming that radon behaved much like xenon, krypton and argon, I rather thought that radon would be appreciably soluble in water via the formation of clathrates, and to some extent this is true. A nice open sourced discussion of radon clathrates and their association with the migration within uranium bearing soils is found here: The origin and detection of spike like anomalies in soil gas radon time series. (Chyi et al Geochemical Journal 25 431-438 (2011))
The idea of radon clathrates suggests that for whatever reason, something rather like hydrogen bonding, radon is hydrophilic.
The behavior of radon in tissue has been studied and a number of interesting papers on the subject have been published, using both animal and human subjects. A very recent paper on the subject explores the theoretical justification for the observed fact that physiologically, radon tends to have an affinity for hydrophobic lipids, that is fat tissue. The paper is also open sourced and is available here: A combined experimental and theoretical study of radon solubility in fat and water (Barbara Drossel et al, Scientific Reports, (2019) 9:10768).
It is now well known that radon exposure can lead to immune suppression. One reason for this is that, because of its affinity and solubility in lipids, one place that the gas tends to concentrate is in bone marrow, which is where blood cells, including immune cells, are formed. While severe immune suppression is dangerous, there are physiological conditions for which some immune suppression is desirable, for example, in the case of autoimmune diseases like lupus or rheumatoid arthritis. (Biologic drugs like Humira - adalimumab - work by moderate immune suppression.) While the fad in the early twentieth century of people staying in caves where radon gas is present in high concentrations for therapeutic benefit seems absurd in modern times, there have been controlled clinical trials in which patients suffering from autoimmune diseases have stayed in radon bearing "spas" to evaluate their effect on disease. Many of the papers reporting on these trials refer to the concentration of radon in bone marrow, for example this paper: Decrease of Markers Related to Bone Erosion in Serum of Patients with Musculoskeletal Disorders after Serial Low-Dose Radon Spa Therapy (Claudia Fournier et al Front. Immunol. 8:882 (2017)). The concentration of radon gas into lipid bearing tissues means that the radon daughters are formed there. Thus in high enough concentrations, it will lead to blood related diseases. Marie Curie, for example, who is reported to have traveled around her lab with vials of glowing radium in her pockets died apparently from aplastic anemia, apparently because radon and its daughters slowly destroyed her bone marrow leading to an inability to generate new red blood cells.
(Interestingly a comprehensive study of the fate of uranium miners among the Dine (Navajo) people found that while rates of leukemia were slightly higher than expected among all uranium miners - 17 of them died from leukemia, whereas the expected number of deaths from leukemia would have been around 15, meaning there were two "extra" deaths - Native Americans did not have a single case of leukemia among them, although they were elevated for a number of other cancers, some blood related. Radon Exposure and Mortality Among White and American Indian Uranium Miners: An Update of the Colorado Plateau Cohort (Mary K. Schubauer-Berigan et al, Am J Epidemiol 2009;169: 718–730)).
When people think about risk, they tend to do in an innumerate way, for example, if we report that some improbable thing is 100% more likely in group A than in group B it does not imply that everyone in group A will experience the improbable thing. Consider this paper on the likelihood of dementia among different classes of women depending on their marital status: Marital Status and Dementia: Evidence from the Health and Retirement Study (Liu et al, The Journals of Gerontology: Series B, gbz087, corrected proof, accessed 9/12/19)
Here, simply put are the "results" of the study from the paper:
This brief excerpt of a longer paper reports, caveats included, that 2.41% of divorced women ultimately over a period of more than a decade, develop dementia. It means that of the subjects in this study, 97.59% of the divorced women didn't develop Dementia. Simplistically, we could - especially if we wished to encourage a stupid[ interpretation of the data and annouce that since "only" 1.67% of married women developed dementia that divorced women are 100*2.41/1.67 =144% more likely to develop dementia than married women. Run through the mill of some barely literate journalism, as in the case of the tuna fish above, we could probably find interpretations of this data that implied that all married women should stay in marriages, possibly even abusive marriages as a way of preventing dementia. The fact is however, that whether a woman has never married, is divorced, married, or cohabiting she is still unlikely to develop dementia.
As I played around with the data on the uranium miners on the Colorado Plateau in the aforementioned paper, I wrote the following elsewhere on the internet in the link on the question of whether uranium is exhaustible:
...Of the Native American miners, 536 died before 1990, and 280 died in the period between 1991and 2005, meaning that in 2005, only 13 survived. Of course, if none of the Native Americans had ever been in a mine of any kind, never mind uranium mines, this would have not rendered them immortal. (Let’s be clear no one writes pathos inspiring books about the Native American miners in the Kayenta or Black Mesa coal mines, both of which were operated on Native American reservations in the same general area as the uranium mines.) Thirty-two of the Native American uranium miners died in car crashes, 8 were murdered, 8 committed suicide, and 10 died from things like falling into a hole, or collision with an “object.” Fifty-four of the Native American uranium miners died from cancers that were not lung cancer. The “Standard Mortality Ratio,” or SMR for this number of cancer deaths that were not lung cancer was 0.85, with the 95% confidence level extending from 0.64 to 1.11. The “Standard Mortality Ratio” is the ratio, of course, the ratio between the number of deaths observed in the study population (in this case Native American Uranium Miners) to the number of deaths that would have been expected in a control population. At an SMR of 0.85, thus 54 deaths is (54/.085) – 54 = -10. Ten fewer Native American uranium miners died from “cancers other than lung cancer” than would have been expected in a population of that size. At the lower 95% confidence limit SMR, 0.64, the number would be 31 fewer deaths from “cancers other than lung cancer,” whereas at the higher limit SMR, 1.11, 5 additional deaths would have been recorded, compared with the general population.
As noted, 63 more uranium miners among the more than 2,400 Native American miners died from lung cancer than would have been expected in a "normal" population. However, more than 2,300 Native Americans, despite all the books about the horrible conspiracy to kill them, did not die from lung cancer. How many more might have died than a similar sized population of school bus drivers in Indianapolis if they hadn't mined uranium but rather just grew up on a huge uranium formation - some of the rocks included the construction of Pueblos made hundreds of years ago show significant radioactivity from the natural composition of the rock, some of which are decent uranium ores - is not known, but it is clear, I think, from all of the above, that exposure to the decay products of uranium is not good for you, even if there are people who deliberately expose themselves to them in hopes of "curing" their rheumatoid arthritis.
However exposure to decay products of uranium does not mean either instantaneous nor even likely death from radon related causes. I have measurable and detectable radon in the room in which I am writing this post. This radon has nothing to do with anthropomorphic activities, but is a function of the natural composition of the soils and rocks under the house in which I have lived. Note, I may get lung cancer from this radon, but it is improbable that I will, just as I may get lung cancer from the air pollution with which I have lived my whole life, much of it while people with selective attention and wishful thinking wait, like Godot, for the grand so called “renewable energy” nirvana that has not come, is not here, and will not come. Even though I definitely have been exposed to radon, I am decidedly not dead, and to my knowledge, do not have lung cancer, although I've lived in this house for more than 23 years. However, a set of people consisting of people living in houses like mine will have a higher probability, but not a certainty, of dying from lung cancer. The difference between “higher probability” and "certainty" is important.
Recently in this space, there was a post on the subject of outcome bias: The bias that can cause catastrophe. This post referred to an argument that an observed outcome is assumed to have been a likely outcome, when this is decidedly not true. It more or less follows to my mind that advertising - including advertising disguised as "news" - can lead to the catastrophic outcomes, for example the bizarre and intellectually unsupportable and frankly extremely dangerous arguments for example that nuclear energy is dangerous unacceptable and climate change and the deaths of millions of people each year from air pollution is acceptable and not dangerous, that nuclear energy is "too expensive" and that the climate change and the deaths of millions of people each year from air pollution is not "too expensive." You can hear moral idiots drag out these arguments time and time again, here and elsewhere; the most odious, ignorant, appalling, weak minding and repulsive examples of such people here have made it to my wonderful ignore list. I don’t listen to what Donald Trump has to say, why listen to other people who are, frankly, delusional. It is a fact that over the last half a century, the combustion associated with so called “renewable energy” – including but not limited to the combustion of “waste” and biomass, will kill more people this year than more than half a century of commercial nuclear operations.
Facts matter.
Because we advertise Fukushima more than we report climate change deaths and air pollution deaths, we have chosen, via outcome bias, to destroy the planet by appeals to fear and ignorance.
All this brings me to the papers, finally, evoked at the outset of this post.
The San Joaquin Valley is primarily an agricultural valley, producing nuts, vegetables - almost all this country's asparagus is grown there - according to Wikipedia diverse crops are grown there: Walnuts, oranges, peaches, garlic, tangerines, tomatoes, kiwis, hay, alfalfa, cotton, pistachios, almonds, and of course oranges.
The overwhelming majority of the State's dangerous petroleum is produced there.
The first paper, that on the radium content of groundwater in California's San Joaquin oil fields, reports oil has been produced there for a century. To wit, from the introduction:
The focus of this study is shallow groundwater associated with the Fruitvale (FV), Lost Hills (LH), and South Belridge (SB) oil fields in the SJV (Figure 1),12 where oil production has occurred for ?100 years.8 Disposal of oil-field water in unlined ponds has occurred in parts of the study area since the 1950s and is a direct pathway for oil-field water to enter the near-surface environment. 13 Several studies have reported the presence of Ra from oil-field water in near-surface environments, typically in aquatic sediment or soil associated with releases of Ra-rich produced water.6,14?16 Those studies found most of the Ra was retained on solid phases relatively close to the release site due to Ra immobilization by processes like coprecipitation with barite (BaSO4) and adsorption on solid phases.6,14,15
The location of these oil fields and the test sites are detailed in this map:

The caption:
The following graphic refers to the findings of total radioactivity in groundwater and oil field water. The "MCL" again, refers to the EPA's "maximum concentration level" for radium, 0.185 Bq/Liter. Several of the groundwater samples exceed this limit, all of the oil field water do as well.

The next three graphics and captions show technical geochemical issues that have to do with the migration of radium as the oil field water leaches into the ground. Since the paper is open sourced, one is free to read the technical discussions therein, if interested.



It is worthwhile to read the text in the original paper. It appears that the radium, at least in some of the oil field produced water, did not migrate directly into the wells, but rather that the salts in this water mobilized radium that was geochemically fixed by manganese that was reduced, and then mobilized to produced the high radium levels. Nevertheless, the groundwater radium concentrations were increased because of oil drilling activity in the area. To the extent that the oil field water dries out, of course, leaving behind dust and oil residues, the Santa Ana winds could always blow this surface radium around across the fields.
The authors' conclusions/implications:
Implications
Chemical and isotopic data from this study show that saline, organic-rich oil-field water infiltrated through unlined disposal ponds into groundwater in multiple locations on the west side of the SJV. In three locations identified in this study, this has induced rock-water interactions that mobilize Ra from downgradient aquifer sediments to groundwater at levels that exceed the 226Ra+228Ra drinking-water MCL. These processes could also control Ra distribution in other areas with surface releases of produced water, rather than assuming high Ra is related to Ra adsorbed to sediment near the release site or that Ra activity in impacted groundwater depends only on conservative mixing relationships between the oil-field water and ambient groundwater. Induced-radium mobilization by oil-field and other saline water sources should be further studied in other cases, even if the end-member saline source has low Ra activity.
Before turning to the paper on the Fukushima seaweed, having already disposed of the famous Fukushima Tuna fish above, it is worth considering the entire amount of Cesium-137 released into the ocean by Fukushima, a favorite topic of anti-nukes, a set, as I often note, of people who are spectacularly disinterested in the fact that 19,000 people will die from air pollution today.
I will take this reference for the total amount of Cesium-137 released into the ocean: Oceanic dispersion of Fukushima-derived Cs-137 simulated by multiple oceanic general circulation models (Kawamura et al Journal of Environmental Radioactivity 180 (2017) 36-58). There surely other papers that will vary in exact figures, but the order of magnitude is surely reliable; in table 3, in the paper it reports that 3.53 petabequerels were released into the ocean.
Anti-nukes are hysterical about radioactivity and one of the more stupid remarks they make is that "There are no 'safe' levels of radioactivity." This bit of sophistic ignorance ignores the fact that there is a minimum amount of radioactivity that one must contain in order to live. According to a well known popular book on science (Emsley, John, The Elements, 3rd ed., Clarendon Press, Oxford, 1998) a 70 kg human being, contains about 140 grams of potassium. All of the potassium on earth is radioactive, owing to the presence of the K-40 isotope, which has a half-life of 1.277 billion years, making its radioactivity appreciable, but it's half-life long enough to have survived in the star explosion debris from which our planet accreted. From the isotopic distribution of natural potassium, one can easily calculate that a 70 kg human being contains about 259,000 Bq of radioactive potassium. Without this radioactivity, a human being would die and die quickly, since potassium is an essential element.
It follows that for a population of 7 billion, the radioactivity of all human beings on earth is roughly 30 trillion Beq, 30 terabequerel.
The half-life of cesium-137 is, again, 30.08 years. It is therefore straight forward, using the radioactive decay equations, to show that the period of time it would take for all the cesium-137 released into the ocean to decay to the same level found in all human beings is 207 years. (For reference, I have calculated that the ocean contains about 20 zetabequerel of potassium, meaning the radioactivity in the ocean exceeds the radioactivity released at Fukushima by a factor of over 5,400,000.) The period of time it would take for all the cesium-137 released into the ocean by Fukushima to decay to amount of cesium in a single 70 kg human being, just one person, is 1013 years.
Since the half-life of radium is 1600 years, this implies that the amount of radium in the oil field water in the San Joaquin Valley will be in 207 years, 91.4% of what it is today; in 1013 years, the amount will be 64.4% of what it is today. This means that oil field radioactivity will be present in significant amounts after roughly the same time period of time that has elapsed since the Battle of Hastings, when France conquered England in 1066.
Of course, the radium that was in the ground in the San Joaquin valley and has now been brought to the surface by oil drilling has been there since the valley formed.
Let's be clear on something too: Claiming that it was wise to bet the planet on so called "renewable energy" is doing nothing to stop this state of affairs, nothing at all. The use of petroleum on this planet grew, in this century, grew by 30.23 exajoules to a total of 185.68 exajoules. So called "renewable energy" as represented by solar and wind, by comparison grew by 8.88 exajoules to 10.63 exajoules. (Total Energy consumption from all forms of energy was 584.98 exajoules in 2017, the year from which this data is taken.) 2018 Edition of the World Energy Outlook Table 1.1 Page 38 (I have converted MTOE in the original table to the SI unit exajoules in this text.) Thus, the insistence that we will someday survive on so called "renewable energy" is de facto acceptance of the use of fossil fuels, acceptance of the 7 million air pollution deaths each year, and acceptance of climate change. All the Trumpian scale lies repeated year after year, decade after decade will not make any of these statements untrue.
Now let's turn to the paper on Fukushima and seaweed.
Here's the cartoon art for the abstract:

This paper reports a similar quantity of cesium-137 as having been released into the ocean as the previous paper, but only as the lower end of a range of 3.5 to 5.5 petabequerels.
From the introduction:
Large earthquakes and an associated tsunami in March 2011 resulted in an accident at the Fukushima Daiichi Nuclear Power Plant (FDNPP). Immediately after the accident, a large amount of radioactive material was released into the atmosphere from the FDNPP, a large portion of which was deposited in the ocean.1?3 In addition, a large amount of radioactive material (e.g., 3.5?5.5 PBq in 137Cs) was released directly from the FDNPP into the ocean, resulting in a high level of radiocesium entering the sea.4?9
Many marine organisms were contaminated by radioactive materials in direct inflow water from the FDNPP; therefore, monitoring of the radionuclide concentration of marine biota was begun.10?13 The main nuclides monitored were radioactive iodine (131I), radiocesium (134Cs and 137Cs), and radioactive silver (110mAg), and high levels of these materials were indeed detected in most marine organisms collected from the Fukushima coast immediately after the accident.10 The levels of radiocesium in fish and invertebrates decreased over time;14,15 however, several independent studies have been conducted to study their concentrations in marine plants since the accident.13,14,16?19
According to the Tokyo Electric Power Company (TEPCO),19 in addition to 134Cs and 137Cs, 110mAg was also detected in invertebrates and marine plants collected during investigation of radioactive materials in coastal marine organisms following the accident. The 110mAg that originated from the FDNPP accident was also detected in other marine organisms and fish;10,11,19 however, reports of these compounds were fragmentary and did not discuss changes in levels after the accident in detail. Marine plants are important primary producers; accordingly, it is important to clarify the concentrations of radionuclides in marine plants and their changes because of the potential for plants to transfer radioactive materials in ecosystems. Therefore, in this study, the temporal changes and behavior of the concentrations of radioactive materials 110mAg, 134Cs, and 137Cs in marine plants were investigated.
Here are the time points and species collected from the methods section:
Sixteen plant species were collected and studied, eight brown algae species, seven red algae and one sea grass, 83 samples for each site. They were washed with artificial seawater and then subject, using normal procedures, to measurement of their radioactive profiles.
The following figure shows where the samples were collected:

The caption:
The authors' discuss the biological half-life of the three radioactive species studies, and their ecological half-life, as opposed to their radioactive half-lives.
The following table from the paper shows the results:

Graphics illustrating the same things.

The caption:
NRA here stands not for an organization of homicidal Trump voters but for the (presumably Japanese) "Nuclear Regulatory Administration."
Organisms can, and often do, concentrate elements from their environment. For example, coral concentrates uranium from seawater, and in fact, corals, were they not about go extinct because of ocean acidification and climate change while we all wait for the so called "renewable energy" nirvana that did not come, is not here, and will not come, would be low grade uranium ores.
This is true of cesium in seawater, as shown in the following graphic.

The caption:
From the text:
Apparent Concentration Factor and Radioactive Nuclide Ratio of Marine Plants.
The 137Cs concentration of seawater after the accident was very high;6,7 however, it decreased rapidly to 0.1 Bq/L approximately 500 days after the accident at both sites. The concentration subsequently fluctuated, until it decreased to ?0.01 Bq/L after 1500 days, which was close to the value before the accident23 (Figure 2d). The 137Cs concentration of seawater in both areas decreased significantly over time (Spearman’s rank correlation coefficient; for Yotsukura, rs = ?0.655 and p < 0.01, and for Ena, rs = ?0.682 and p < 0.01).
The temporal changes in the 137Cs concentrations of marine plants corresponded well with those of seawater. An apparent concentration factor (ACF) was calculated from the ratio of the 137Cs concentration of marine plants and seawater off Yotsukura and Ena. Samples that were below the detection limit were removed from the analysis.
Temporal variations in the ACF of 137Cs of P. iwatensis and E. bicyclis are shown in Figure 3. The ACF of each species was 2.90?244 for P. iwatensis and 4.20?192 for E. bicyclis. The ACF values of all samples increased until day 593 (October 2012) and then decreased until day 1041 (January 2014), after which they again showed a tendency to increase. The temporal changes in 134Cs/137Cs activity ratios and the 110mAg/137Cs activity ratios are shown in Figure 4.
Figure 4:

The caption:
From the paper's conclusion:
In this study, the lower detection limit was ?0.1 Bq/kg of WW. However, since 2015, no 110mAg has been detected in P. iwatensis and E. bicyclis, and radioactive Cs has been detected in only a few marine plant samples, indicating that the transfer of radionuclides to the ecosystem in the future will be extremely small.
None of this implies that the Fukushima accident was not serious, although the majority of deaths in the area had nothing to do with nuclear power; the deadliest feature of the tsunami was seawater, and the argument that nuclear power should be abandoned because of Fukushima represents highly selective attention. If radiation from the destroyed reactors makes them "too dangerous" it follows that living in coastal cities is too dangerous, since almost all of the deaths connected with the event involved seawater and not radiation.
Air pollution kills seven million people each year. Since March of 2011, close to 60 million people have died from air pollution, roughly half the population of Japan.
Radionuclides have been and are released into the environment by the use of commercial nuclear power, but relative to natural radioactivity, the amounts are very small and the relative risk is small.
I always know I'm speaking to a fool when - and this happens a lot - the subject of the nuclear weapons plant at Hanford, where radioactive materials are migrating from leaking tanks, comes up when nuclear energy comes up. There was actually an ass here who called up one of my old posts to point out how "dangerous" nuclear power was (at least in his withered mind) because a tunnel on the Hanford reservation, which contained apparently some old rail cars on which decommissioned chemical reactors (with plutonium and other nuclide residues) were placed collapsed. The death toll from this event was zero. Nineteen thousand people, again and again and again, will die today from air pollution, and we have an asshole burning electricity, almost certainly generated using fossil fuels, to complain about the "danger" of some disused chemical reactors.
It boggles the mind that anyone could be so asinine as to think this way.
The technology utilized at Hanford in the creation of the tanks was conducted in an atmosphere of secrecy and war paranoia utilizing technology largely developed in the mid 1940's and 1950's. It was designed to extract weapons grade plutonium, a process which is inherently dirtier than reprocessing commercial nuclear fuels since it is necessary that this plutonium be in very low concentrations in the fuel. It is, in this sense, irrelevant to modern times.
It is almost impossible to grasp how much more chemistry we know than we knew in 1970. In 1970, for example, lanthanides, which are fission products, were largely chemical curiosities. Today they are essential components of modern technology, including many of the so called "renewable technologies" that have proved to be unsuccessful and overly lauded fetishes, given that they have not arrested the use of dangerous fossil fuels.
Hundreds of billions of dollars are being spent to "clean up" Hanford, while we are not spending hundreds of billions of dollars to provide even a primitive level of sanitation to the more than one billion people on this planet who lack it. Hundreds of thousands people die from fecal waste as a result. There is no evidence that anyone has ever died from the leaching of the Hanford tanks, although perhaps some will someday, but even in Richland, Washington, the closest city to Hanford, it is very, very, very unlikely that the death toll from radioactivity at Hanford will even remotely approach the death toll from eating fatty food in that city, or for that matter, the death toll associated with automobile and diesel exhaust generated from pizza delivery cars and trucks delivering merchandise from China to Walmart.
Now. Some very good research is being done at PNNL, (Pacific Northwest National Laboratory) into the behavior of radioactive materials both in the environment and in processing. Not all of this money being spent there is therefore wasted, even though very few lives, face or will face significant risk as a result of the leaking tanks. If nothing were done at Hanford, many of the nuclides (not all, but many) would decay before the groundwater in them reached the Columbia River. If they do reach the river, they will have been diluted by the migration and finally by the river water, be constrained in sediments and more than likely never appear in concentrations in human flesh in an amount comparable to potassium in flesh.
Let's be clear. The renewable nirvana is not here. It's not coming. The insistence that we shut nuclear plants because people have an inordinately paranoid reaction to radioactivity because they are, frankly, extremely poorly educated, is killing the planet and the rate at which it is dying is accelerating, not slowing. It is not close to slowing.
Unless we think clearly, all of this is going to get worse, and, besides a destroyed atmosphere, we will have begun to mobilize radium and dump it on the surface of the planet where it will remain for thousands of years in a completely uncontrolled fashion. The radium, however, is nothing like the risk of carbon dioxide and climate change and the only reason to reflect upon it is to point out the hypocrisy of anti-nukes, who accept oil and gas radiation but are unwilling to accept the far less risky radiation in contained nuclear fuels.
Well, I'm having a wonderful afternoon, since I'm finally getting this post out. It's not perfect, I'm sure, but I certainly learned some interesting things for writing it. It took a long time, and I abandoned it several times, but this is a point I wanted to turn over in my mind, even if no one reads or likes it.
If you are reading it, I trust your day will as pleasant as mine is. Have a great weekend.
Profile Information
Gender: MaleCurrent location: New Jersey
Member since: 2002
Number of posts: 33,515